Voltaic Cells
This guide delves into essentials of electrochemical setups, including various battery types like dry cells, lithium-ion, and lead-acid batteries. It explains how these cells generate electricity through spontaneous reactions, with a focus on their voltage, chemical reactions, and energy storage capabilities. Insight into fuel cells and their reactions is also provided. Learn about the construction and chemical processes behind lead-acid batteries, their applications in automobiles, and how they deliver substantial power for quick energy needs.

Voltaic Cells
E N D
Presentation Transcript
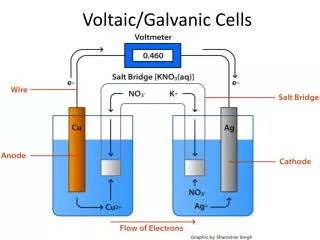
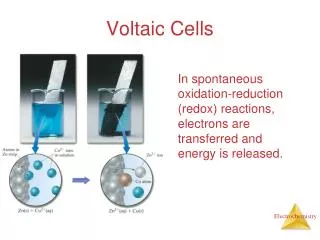
Voltaic Cells Batteries, etc.
Essentials • Electrochemical setups that can generateelectricity • They release energy and are spontaneous • Ecell is positive (by definition) • Viewed as sources of energy • Voltage depends on the chemical reaction • Amount (amps) depends on the size of the cell
Many types of batteries • Dry Cells ("D", "AA" "AAA" ) • Lithium ion battery • Car Batteries (Lead Acid) • Rechargable (can be some types of lithium or Nickel/ Cadmium (Ni/Cad) • Fuel Cells (Hydrogen/Oxygen)
The Chemical Reaction Overall 2MnO2 + Zn Zn2+O + Mn2O3 The zinc is oxidized: Zn0 Zn2+ + 2 e- The manganese is reduced: 2 x [1 e- + (Mn4+)O2 ] (Mn3+) 2O3
The Lead Acid Battery Used in automobiles Made of Lead plate, lead oxide and sulfuric acid nasty and toxic Great for holding a LOT of electricity Good for rapid charge and discharge Lots of rapidly available power (as in turning around a 300 pound car engine fast enough to get it started) Made of 6 cells put together: each cell gives 2 volts, Together they make 12 volts.
The chemical reaction: 2 e- The discharge… PbO2 + 2 H2SO4 + Pb2PbSO4 + H2O Some lead is oxidized: Pb0 Pb4+ + 2 e – (SO4 2- is a spectator) Some lead is reduced: 2 e - + Pb4+O2 Pb4+ + H2O (SO4 2- is a spectator)