Understanding the Neutralization of Acids and Bases: Key Concepts and Reactions
This guide provides a comprehensive overview of the neutralization of acids and bases, focusing on titration curves and pH changes during a neutralization reaction. It explores the definitions of acids and bases, details acid-base reactions, and offers examples of common oxides and their classifications. Key reactions and properties of strong bases, acidic oxides, and amphoteric oxides are discussed, providing a broad understanding of their roles in chemical processes. The information serves as an educational resource for students and chemistry enthusiasts.

Understanding the Neutralization of Acids and Bases: Key Concepts and Reactions
E N D
Presentation Transcript
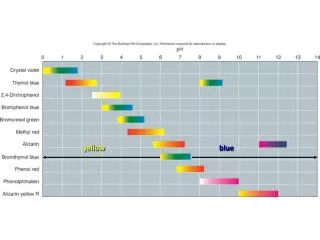
yellow blue
Titration Curve of a Neutralisation Reaction pH of Acid solution starts off very low SA Base is being added; pH rises
Definitions Acids: Eg. HCl(g) H+(aq) + Cl-(aq) A substance when in solution produces hydronium ions (H3O+); proton DONOR
Acids Bases Bases:
Definitions Bases: Proton ACCEPTOR A substance which contains a hydroxide ion (OH-) or oxide (O2-) A substance when in solution produces hydroxide ions (OH-)
Acid – Base Reactions Neutralisation Reaction occurs when acids and bases react to form ionic salts ACID + BASE WATER + SALT HCl(aq) + NaOH(aq) H2SO4(aq) + K2O (aq) HBr(aq) + NH3(aq)
Acid or Base? a) ClO2 ...... ___________ f) CO2 ...... ___________ b) SrO...... ___________ g) Cs2O ... ___________ c) Cr2O3 ... ___________ h) BaO ..... __________ d) SeO2 .... ___________ i) SO3 ...... ___________ e) NO2 ..... ___________ j) Ag2O ..... ___________ What do these molecules all have in common??
Acids Bases Which of those compounds would produce oxide ions??
Basic Oxides • Are “metal” oxides • Eg. Na2O, K2O, MgO, CaO, Fe2O3, Ag2O….. • They all dissociate to produce O2- ions which acts as a STRONG BASE Eg. Step 1: K2O K+ + O2- Step 2: O2- + H2O 2OH-
Acidic Oxides • “Non-metal” oxides eg. CO2, NO2, P2O3, SO3…. • Remains covalently bonded to oxygen • Chemically combines with water to form an acidic solution eg. SO2 (g) + H2O H2SO4 Eg. SO2
Amphoteric Oxides Certain metals and metalloid oxides can act as either an acid or a base: Eg. Al2O3+ 6HCl → 2AlCl3 + 3H2O Base Acid Al2O3+ 2NaOH → Na2Al2O4 + H2O Acid Base