Molecular Geometry and Bond Polarity in Chemistry
280 likes | 309 Vues
Explore VSEPR theory to determine molecular shapes & polarity. Learn about bond angles, types of electron pairs, and bond polarities using examples and electronegativity trends. Understand the difference between nonpolar and polar molecules.

Molecular Geometry and Bond Polarity in Chemistry
E N D
Presentation Transcript
Ch. 9 – Molecular Structure II. Molecular Geometry
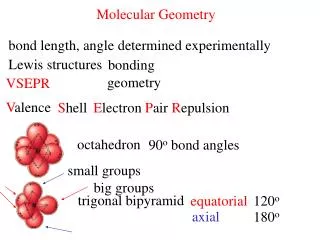
A. VSEPR Theory • Valence Shell Electron Pair Repulsion Theory • Electron pairs orient themselves in order to minimize repulsive forces.
Lone pairs repel more strongly than bonding pairs!!! A. VSEPR Theory • Types of e- Pairs • Bonding pairs - form bonds • Lone pairs - nonbonding e-
Bond Angle A. VSEPR Theory • Lone pairs reduce the bond angle between atoms.
Know the common shapes & their bond angles! B. Determining Molecular Shape • Draw the Lewis Diagram. • Tally up e- pairs on central atom. • double/triple bonds = ONE pair • Shape is determined by the # of bonding pairs and lone pairs.
BeH2 C. Common Molecular Shapes 2 total 2 bond 0 lone LINEAR 180°
BF3 C. Common Molecular Shapes 3 total 3 bond 0 lone TRIGONAL PLANAR 120°
SO2 C. Common Molecular Shapes 3 total 2 bond 1 lone BENT 104.5°
CH4 C. Common Molecular Shapes 4 total 4 bond 0 lone TETRAHEDRAL 109.5°
NH3 C. Common Molecular Shapes 4 total 3 bond 1 lone TRIGONAL PYRAMIDAL 107°
H2O C. Common Molecular Shapes 4 total 2 bond 2 lone BENT 104.5°
PCl5 C. Common Molecular Shapes 5 total 5 bond 0 lone TRIGONAL BIPYRAMIDAL 120°/90°
SF6 C. Common Molecular Shapes 6 total 6 bond 0 lone OCTAHEDRAL 90°
F P F F D. Examples • PF3 4 total 3 bond 1 lone TRIGONAL PYRAMIDAL 107°
OCO D. Examples • CO2 2 total 2 bond 0 lone LINEAR 180°
C. Bond Polarity • Electronegativity Trend • Increases up and to the right.
+ - H Cl A. Dipole Moment • Direction of the polar bond in a molecule. • Arrow points toward the more e-neg atom.
B. Determining Molecular Polarity • Depends on: • dipole moments • molecular shape
F BF3 B F F B. Determining Molecular Polarity • Nonpolar Molecules • Dipole moments are symmetrical and cancel out.
O net dipole moment H2O H H B. Determining Molecular Polarity • Polar Molecules • Dipole moments are asymmetrical and don’t cancel .
H net dipole moment CHCl3 Cl Cl Cl B. Determining Molecular Polarity • Therefore, polar molecules have... • asymmetrical shape (lone pairs) or • asymmetrical atoms
C. Bond Polarity • Most bonds are a blend of ionic and covalent characteristics. • Difference in electronegativity determines bond type.
EN Difference • 0.0 - 0.4 Nonpolar • 0.4 - 1.0 Moderately Polar Covalent • 1.0 - 2.0 Very Polar Covalent • > 2.0 Ionic
C. Bond Polarity Examples: • Cl2 • HCl • NaCl 3.0-3.0=0.0 Nonpolar 3.0-2.1=0.9 Moderately Polar Covalent 3.0-0.9=2.1 Ionic
C. Bond Polarity • Electronegativity • Attraction an atom has for a shared pair of electrons. • higher e-neg atom - • lower e-neg atom + ____
C. Bond Polarity • Nonpolar Covalent Bond • e- are shared equally • symmetrical e- density • usually identical atoms
- + C. Bond Polarity • Polar Covalent Bond • e- are shared unequally • asymmetrical e- density • results in partial charges (dipole)