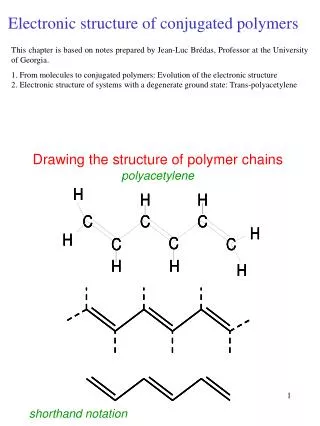
Chemical Idea 2.3: Electronic structure
140 likes | 321 Vues
Chemical Idea 2.3: Electronic structure. Learning objectives Describe the Bohr model of the atom Write electronic configurations for the first 36 elements Relate shape of the periodic table to electronic structure. Rutherford’s Atom. The problem with Rutherford’s atom was the electrons…..

Chemical Idea 2.3: Electronic structure
E N D
Presentation Transcript
Chemical Idea 2.3: Electronic structure Learning objectives Describe the Bohr model of the atom Write electronic configurations for the first 36 elements Relate shape of the periodic table to electronic structure
Rutherford’s Atom • The problem with Rutherford’s atom was the electrons….. • …… no one could understand why they did not collapse into the nucleus. • The model of the atom was still based on classical physics. Bohr was one of the first scientists to try to use Einstein’s new ideas about quantum theory, and quanta discrete packets of energy, with certain fixed values.
Bohr’s Atom • Bohr studied the hydrogen emission spectrum and found that it was made up of a number of discrete lines, in the visible region…..
Bohr’s idea • Bohr decided that each line must be caused by the electron moving between different energy levels. • When an electron is given energy (excited) it can jump into a higher energy level. Later it falls back down again and gives out (emits) the extra energy as light
Controversial! • Quantum mechanics was not popular with scientists because it seemed to contradict a lot of their ideas! • So Bohr’s ideas were not warmly welcomed as it lent a lot of support to the ideas of quantum theory.
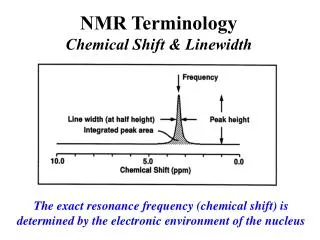
Key Points • The electron in the hydrogen atom could only have certain fixed values. • A photon (particle of light) is emitted or absorbed when the electron moves from one level to another. • Energy of the photon equal to the difference in energy of the 2 shells • ΔE=hν (frequency of photon related to energy difference)
Each line represents a ‘transition’ from a higher ‘excited’ level to the groundstate (lowest) After a certain amount of energy has been added (where shells converge) the electron becomes separated, or ionised. Spectral lines get closer together towards the high energy end- shells become closer (attraction to + nucleus limits distance) Each ring represents an ‘energy level’ or ‘shell’ The lowest level is closest to the nucleus.
Bohr’s Hydrogen atom • Bohr assigned each shell a special number the Principle Quantum Number (n). • He found a special rule about how many electrons could fit into each shell. • Number of e- = 2n2 • For shell 1, 2x 12 = 2 (first shell holds 2 e-) • For shell 2, 2x 22 = 8 (second shell holds 8 e-) • For shell 3, 2x 32 = 18 (third shell holds 18 e-)
Hang on a minute….. • That’s not quite what we learnt at GCSE…… • At GCSE we stop thinking about electrons at Calcium, because that’s where it all starts to go wonky!!
Shape of PT continued… • Row 1, shell 1, 2e- • Row 2, shell 2, 8e- • Row 3, shell 3, 3e- • We were happy with this at GCSE but there’s something not quite right now…….
We have to fill up the lowest energy level first. • There is an overlap between the 4th and 3rd energy levels • We need to sneak 2 electrons into the forth shell before we continue to fill up the third shell • so the table goes wonky! (transition metals) • The same thing happens for the Actinides and Lanthanides. • because most of them are short lived and radioactive, chemists don’t use them very much • we move them to the bottom, or chop them off completely!!