Periodic Table and Configuration
E N D
Presentation Transcript
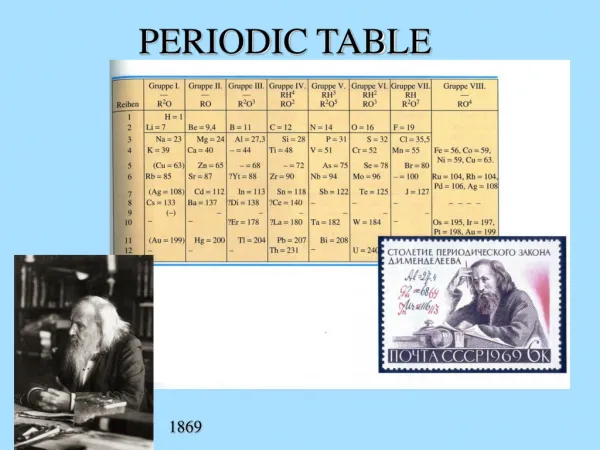
Demetri Mendeleev • Created modern periodic table (late 1800’s) • Arranged by increasing atomic mass • Similar elements found in columns
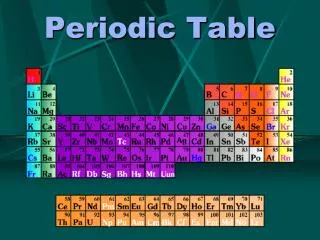
Modern Table • Elements are in columns called groups or families • All elements within a group have similar chemical properties and can often be used to replace one another in compounds to produce new compounds with slightly different properties • Horizontal rows are called periods
Three Distinct Regions • Representative elements are chemically active elements • Transitional metals are in center of the table • Inner transitional metals are the two rows below 1) top row called lanthanides 2) bottom row called actinides
Group Names • Group 1 – Alkali Metals • Group 2 – Alkaline Earth Metals • Group 16 – Chalcogens • Group 17 – Halogens • Group 18 – Noble or Inert Gases
Metallic Character • Elements can also be divided by metallic character • Metalloid – between metal and non-metal
Configuration • Arrangement of electrons in the atom • Aufbau Principle – orbitals fill in order of increasing energy
Orbital Diagram of Li ( 3 e-) • 1s22s1
Orbital Diagram of B (5 e-) • 1s22s22p1
Hund’s Rule • Electrons occupy degenerate orbitals singly first
Orbital Diagram of C (6 e-) • 1s22s22p2
Orbital Diagram of Ne (10 e-) • Configuration 1s22s22p6
Example • Determine the configuration of Fe Br
Magnetism • When electrons in half-filled degenerate orbitals spin in the same direction, the material is magnetic
Valence Electrons • Outer shell electrons • Ones in the last main energy level ▪ K 1s22s22p63s23p64s1 1 valence e • Fe 1s22s22p63s23p64s23d6 2 valence e