Molecular Geometry VSEPR Theory
Molecular Geometry VSEPR Theory. Section 12.3. VSEPR. Valence Shell Electron Pair Repulsion Electron pairs around central atom arrange themselves to be far apart as geometrically possible. Good help sites: http://www.chem.purdue.edu/gchelp/vsepr/


Molecular Geometry VSEPR Theory
E N D
Presentation Transcript
Molecular Geometry VSEPR Theory Section 12.3
VSEPR • Valence Shell Electron Pair Repulsion • Electron pairs around central atom arrange themselves to be far apart as geometrically possible. • Good help sites: http://www.chem.purdue.edu/gchelp/vsepr/ • http://www.classzone.com/cz/books/woc_07/resources/htmls/ani_chem/chem_flash/popup.html?layer=act&src=qtiwf_act065.1.xml
Linear • 2 things • Bond Angle = 180o • Ex: CO2 180o
3 things Bond Angle = 120 Ex: BCl3 Trigonal Planar 120o
H C H H H Tetrahedral • 4 things • Bond Angle = 109.5 • Ex: CH4 109.5o
Unshared e- Unshared pairs of electrons are extremely close to the central atom compared to atoms forming bonds All the “things” are in the same orientation around central atom, but the only “things” we see in the molecule are the atoms forming bonds
Unshared e- (continued) N H H H 107o • For example: NH3 • Draw the electron dot structure • Determine how many “things” and also the shape based on “things”?
Unshared e- (continued) Therefore with NH3 we expect to see this because it has four things: But one of these spheres would be replaced by a pair of e- REALLY close to the central atom, so we actually see this:
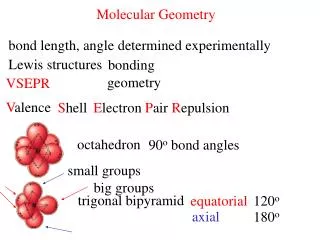
Trigonal bipyramidal • 5 things = trigonal bipyramidal • Bond angles: (90, 120, 180) • Ex.: SF5
Octahedral • 6 things • Bond angles: (90 ,180) • Ex.:PF6
Steps: • Draw electron dot or Lewis structure for molecule/ion. • Count “things” around central atom. • bonds (single, double, or triple each just count as one) • non-bonding pairs (aka unshared pairs, lone pairs) • These “things” are more technically called “regions of electron density”
Steps cont.: • Name the arrangement (5 possibilities) • ThingsArrangement • 2 linear • 3 trigonal planar • 4 tetrahedral • 5 trigonal bipyramidal • 6 octahedral
Steps cont.: • a.If the central atom has no unshared pairs, this is the name of the geometry. • Ex: CH4 has 4 “things” so its tetrahedral b. If the central atom has unshared pairs, arrange them as far apart as possible. • Doesn’t matter for 2, 3, or 4 “things” because of symmetry • When 5 things, lone pairs go on equatorial positions first (since they are farther away from other stuff) • When 6 things, lone pairs go across from each other. • Name the molecular geometry based on the bonds.