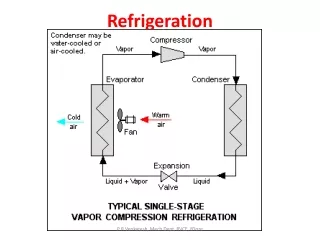
HVAC317 - Refrigeration
HVAC317 - Refrigeration. Refrigeration Theory. Terms. Heat: A form of energy . Refrigeration: The process of removing heat from a space. British Thermal Unit (Btu): The conventional unit of measurement of heat. Terms.

HVAC317 - Refrigeration
E N D
Presentation Transcript
HVAC317 - Refrigeration Refrigeration Theory
Terms • Heat: A form of energy. • Refrigeration: The process of removing heat from a space. • British Thermal Unit (Btu): The conventional unit of measurement of heat.
Terms • A Btu is defined as the amount of heat required to raise or lower the temperature of one pound of water (or any substance) one degree Fahrenheit. • Substances other than water require different amounts of Btu.
Terms • Thermodynamics: The laws of thermodynamics state that heat will always travel from warmer to cooler areas. • Pressure: Standard atmospheric pressure is 14.7 psia (pounds per square inch absolute pressure).
Terms • Refrigeration gauges are calibrated to read zero psig (pounds per square inch gauge pressure) at standard atmospheric pressure. • Vacuum: Pressures below atmospheric pressure.
Terms • Compound Gauge: A gauge that can read above and below atmospheric pressure (this is always the low side gauge on your manifolds). • Above atmospheric pressure these gauges will read psig. • Below atmospheric pressure these gauges will read inches of mercury (Hg).
Terms • Sensible Heat: A change in temperature measured with an ordinary thermometer. • Latent Heat: Heat that causes a change in state. • Change in State: Conversion of a substance from one form to another. This is NOT a temperature change, it is a molecular structure change. • For example: • Water to Ice, or Ice to Water • Water to Steam, or Steam to Water
Terms • Specific Heat: The amount of Btu it takes to raise the temperature of one pound (1 lb) of any substance one degree (1) • Ambient Temperature: The temperature of air that is around an object or device.
Terms • Enthalpy: The total amount of heat that is around a substance. • Ton: A refrigeration rating that is calculated when Btu are known. • 1 ton = 12,000 Btu per hour • Compressor tonnage and horsepower are proportionally equal. • 1 ton = 1 hp = 12,000 Btu/h
Terms • Temperature Pressure Chart (TP Chart): A cross reference between the temperatures, pressures and the boiling points of refrigerant.
Terms • Methods of heat transfer • Convection: Heat transfer by fluid or air movement. (Air Conditioning / Forced air systems) • Conduction: Particle to Particle transfer of heat. (Radiant Heat) • Radiation: Transfer of heat by rays. (sunlight)
Classification of Systems • High Temperature • Start at about 45°F • End at about 60°F • Used in flower shops and candy storage.
Classification of Systems • Medium Temperature • Start at about 30°F • End at about 45°F • Used in products such as dairy goods, beer, wine. • Many products will not freeze at 32°F.
Classification of Systems • Low Temperature Systems • Start at 32°F, and decrease from there • Used in anything frozen such as ice, ice cream, frozen foods, meat storage. • Must have a defrost cycle.
Temp / Pressure Relationship • The temperature at which a substance changes state. It is dependent on pressure. • Lowering the pressure lowers the saturation or boiling point. • Raising the pressure raises the saturation or boiling point.
Refrigeration Calculations • To find the total Btu needed to cool a substance from one temperature to another (without a change in state): • Find the net weight of the substance. • Find the degrees of temperature change (Delta T or T). • Find the specific heat of the substance. • Btu/hr = weight * T * Spec. Heat
Example 1 • Cooling 500 lbs of Cranberries from 70F to 35F T = 70 - 35 = 35 Btu/hr = weight * T * Spec. Heat Btu/hr = 500 * 35 * .91 Btu/hr = 15,925
Example 2 A grocery store takes in 400 lbsof cucumbers and needs to cool them from the 93°F truck to the long term storage temperature of 45°F. Calculate the Btu. T = .93 - 45 = 48 Specific Heat of Cucumbers = .93
Example 3 Need to chill 50 gallons of water from 75°F degrees to 35°F. • Specific heat water = 1.0 • T = 75 - 35 = 40 • 1 gallon water = 8.345 lbs • Weight = 8.345 * 50 = 417.25
Example 3, cont’d: So, it takes 16,690 Btu to cool 50 gallons of water from 75°F to 35°F. The only substance with a specific heat of 1.0 is water.
Refrigeration Calculations • To find the total Btu needed to cool a substance from one temperature to another with a change of state: • Find the net weight of the substance. • Find the degree of temperature change (delta T or T). • Find the specific heat of the substance. • Find the latent heat of the substance.
Refrigeration Calculations • Find the temperature at which the substance changes state (32°F for water). • Calculate the number of Btu prior to the change in state. • Calculate the numer of Btu after the change in state. • Calculate the number of Btu for the state change. • Add them all together for total Btu needed.
Example 1 • A warehouse gets 100 pounds of celery. It is delivered at 70°F. It needs to be cooled to -10°F for long term storage. How many Btu are needed?
Example 1, cont’d • Find the latent heat changes: • Celery freezes at 29.7° F 70 Sensible 29.7 29.7 Latent -10
Example 1, cont’d • Two areas of sensible heat change: • 70 - 29.7 degrees = 40.3°F • 29.7 - -10 degrees = 39.7°F • Latent Heat = 135 Btu
Example 1, cont’d Conclusion: It takes 19,234.1 Btu to cool celery from 70°F to -10°F.
Example 1, cont’d • Note that some substances have a different sensible heat above and below their freezing points.