Balancing Chemical Equations and Calculating Reactant Masses
Learn how to balance chemical equations and calculate reactant masses step by step with examples and atomic mass calculations.

Balancing Chemical Equations and Calculating Reactant Masses
E N D
Presentation Transcript
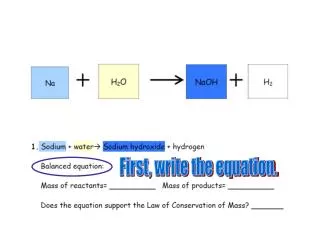
Count the number of atoms on each side of the equation. 1+2=3 Na - _____ H - _____ O - _____ 1 Na - _____ H - _____ O - _____ 1 2 3 1 1
Count the number of atoms on each side of the equation. 2X2=4 2X1=2 2 Na - _____ H - _____ O - _____ 1 Na - _____ H - _____ O - _____ 1 = 2 4 3 1 2 1 =
Count the number of atoms on each side of the equation. 2+2=4 2 2 2 Na - _____ H - _____ O - _____ 1 Na - _____ H - _____ O - _____ 1 = 2 4 3 4 1 2 1 2 =
Count the number of atoms on each side of the equation. 2 2 2 Na - _____ H - _____ O - _____ 1 = Na - _____ H - _____ O - _____ 1 2 4 3 4 = 1 2 1 2 =
Count the number of atoms on each side of the equation. 2 2 2 2 Na - _____ H - _____ O - _____ 1 2 = Na - _____ H - _____ O - _____ 1 2 4 3 4 = 1 2 1 2 =
To find the mass of the reactants: • Find each element on the Periodic Chart • Find the Atomic Mass for each element • Add the atomic mass for each atom to get the total mass for the reactants
46 Na (sodium) has an atomic mass of 23. There are 2 Na (sodium) atoms. 2x23= 46
2x2=4 46 4 H (hydrogen) has an atomic mass of 1. There are 4 H (hydrogen) atoms. 4x1-4
46 4 +32 2x16=32 O (oxygen) has an atomic mass of 16. There are 2 O (oxygen) atoms. 2x16=32
46 4 +32 46 + 4 +32 = 82
46 Na (sodium) has an atomic mass of 23. There are 2 Na (sodium) atoms. 2x23= 46
46 +32 O (oxygen) has an atomic mass of 16. There are 2 Na (sodium) atoms. 2x16= 32
46 +32 +2 H (hydrogen) has an atomic mass of 1. There are 2 H (hydrogen) atoms. 2x1= 2
46 +32 +2 +2 H (hydrogen) has an atomic mass of 1. There are 2 H (hydrogen) atoms. 2x1= 2
46 +32 +2 +2 46+32+2+2=82
82 82 YES