Naming Organic Functional Groups: Alcohols, Carboxylic Acids, Ethers, Aldehydes, Ketones, Esters
E N D
Presentation Transcript
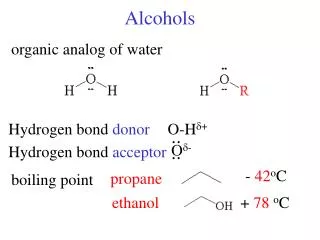
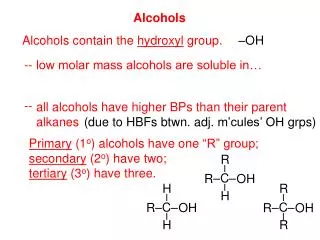
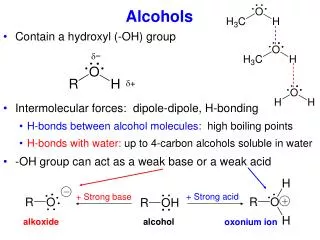
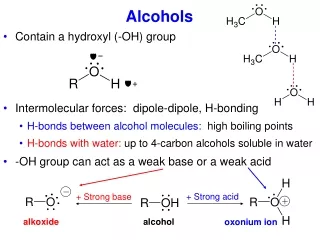
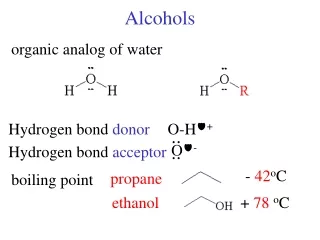
Alcohols • Group: -OH (hydroxyl) on one of the carbons • Naming rules: • Drop e add –ol • Place the number of carbon the OH is attached to in front of the name.
Carboxylic acids • Group: R-COOH or terminal carboxyl group, acts as an acid because • Naming: Drop e and –oic acid.
Ethers • Group: R-O-R or _____________ oxygen between 2 carbon chains • Naming: • Name the two branches in alpha order add ether at end
aldehydes • Group: R-COH or __________________ • Terminal carbonyl Group • Naming: • Drop e add –al
Ketones • Group: R-CO-R or ________________ • Middle of chain carbonyl group • Naming rules: • Use a Number to indicate the carbon that has carbonyl group • Drop e, add -one
Esters • Group: R-COO-R or ___________ • Middle of the chain carboxyl group • Naming Rules • Split molecule in 2 • Put branch name first • For the carboxyl containing end, count carbons to get prefix and then end in oate. (Part of molecule with the double bonded oxygen attached)
Alkanes, Branching Alkanes and Isomers A is butane B is 2-methylpropane Both are isomers of each other. What is an isomer? Molecules with the same molecular formula but different structures/names Picture retrieved 7/26/2019 from: https://www.nyu.edu/classes/tuckerman/adv.chem/lectures/lecture_17/node4.html