Periodic Table
This guide delves into the quantum mechanical model of atoms, focusing on hydrogen's Bohr model and the derivation of quantum numbers. It covers the principal quantum number (n), orbital angular momentum (l), and magnetic quantum number (ml), elucidating how these define electron configurations. The discussion includes the Pauli exclusion principle, the Zeeman effect, and how electrons fill energy levels according to specific rules. With a detailed example of Germanium’s electron configuration, this resource is perfect for students seeking to grasp the fundamentals of quantum chemistry.

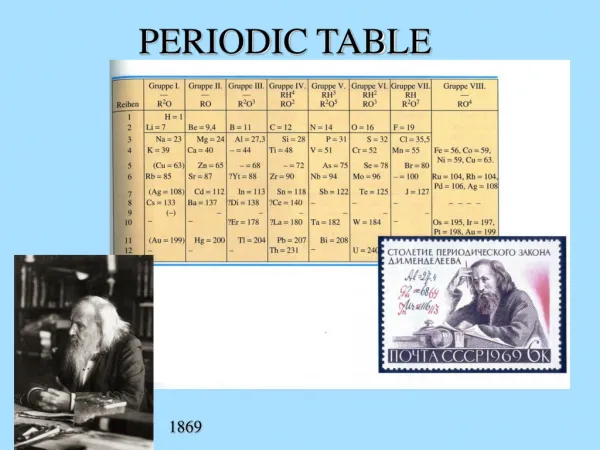
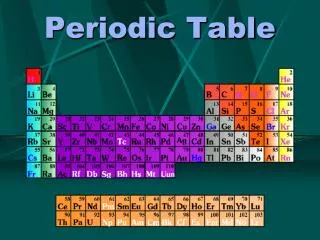
Periodic Table
E N D
Presentation Transcript
The Bohr model of hydrogen provided a single quantum number n. Principal quantum number One-dimensional solution Schrödinger’s equation for a real atom is 3-D. Two other quantum numbers Orbital angular momentum Orbital magnetic number Quantum Numbers 1 2 3 Shells for the principal quantum number n
The orbital angular momentum l is quantized in steps of h. Values from 0 to n-1 The values are named based on early spectroscopy. n shells 1=K, 2=L, 3=M, 4=N, 5=O l shells 0=s, 1=p, 2=d, 3=f, 4=g Orbital Shells A. Clark, Chemical Computing Group
Spectral lines from atoms in a strong magnetic field are split. Zeeman effect Magnetic moment of orbiting electron Orbital magnetic number ml lines up with applied field. Values from –l to l Lz is with respect to field Zeeman Effect
Electrons possess intrinsic angular momentum. Spin angular momentum s Spin magnetic number ms Electron spin s can only have one value: 1/2. ms = +1/2 or -1/2 Spin up or spin down No two atomic electrons can occupy the same set of quantum numbers. Pauli exclusion principle Applies to fermions Photons are spin 1 Spin
Electron Configurations • Electrons in atoms populate the lowest energies first. • Two electrons per state for spin • Atoms have unique ground states. • Hydrogen has 1 1s electron • Helium adds another 1s • Lithium must start 2s shell • Boron starts 2p shell which holds 6 electrons H He Li Be B C N O
Germanium (Ge) has an atomic number of 32. Find the shell configuration for the ground state. Start counting shells from the lowest state. n = 1, l = 0: 1s – 2 electrons n = 2, l = 0: 2s – 2 electrons n = 2, l = 1: 2p – 6 electrons n = 3, l = 0: 3s – 2 electrons n = 3, l = 1: 3p – 6 electrons n = 3, l = 2: 3d – 10 electrons n = 4, l = 0: 4s – 2 electrons n = 4, l = 1: 4p – 2 electrons Total 2+2+6+2+6+10+2+2=32 Germanium
Organized Table • The periodic table was organized to explain chemical properties. • Quantum physics explained structure
Subshell: S G F D P Period 1 1s 2 2s 2p 3 3s 3p 4 4s 3d 4p 5 5s 4d 5p 6 6s 4f 5d 6p 7 7s 5f 6d 7p 8 8s 5g 6f 7d 8p The shells don’t fill up in exact order. Energy levels are determined by quantum calculations. General patterns exist Energy Levels next