Solutions: Solubility, Dissolving, Concentration
450 likes | 470 Vues
Explore the world of solutions, solubility rules, factors affecting solubility, concentration measurements, colligative properties, and heterogeneous mixtures. Learn about acids, bases, and salts as electrolytes and their characteristics. Enhance your knowledge in chemistry.

Solutions: Solubility, Dissolving, Concentration
E N D
Presentation Transcript
Solutions/Acid & Bases Chapter 15 & 19
Solution are homogenous mixtures. • Solutions are made up of 2 things: • A solute & a solvent. • Water is the most common solvent in liquid solutions.
Dissolves in a solvent Example: Sugar in water Will not dissolve in a solvent Example: Sand is insoluble in water. Soluble & Insoluble
Two liquids that are soluble in one another Example: Antifreeze & water Acetic acid & water Two liquids not soluble in one another Example: Oil & vinegar Oil & water Miscible & Immiscible
What gets dissolved. The lesser amount of substance in a solution. Example: Sugar Salt What things dissolve in. The greater amount of substance in a solution. Example: Water Ethanol Solute & Solvent
Solvation • Rules: • Like dissolves like. • Polar dissolves polar. • Nonpolar dissolves nonpolar.
Solvation of salt cont… • Formula units break apart & ions become attracted to charges of the water molecules.
Solvation of sugar (covalent) • In polar molecules, water molecules have a stronger attraction to the polar molecules than they have to each other.
“What can affect solubility?” Two factors that affect rates of solutions. • Temperature • Pressure
Surrounding solute particles with solvent particles. Breaking apart. “Dissolving” Hydration Particles coming together. Dehydration Solvation & Crystallization
Max amount of solute dissolves in a solvent at a temp/pres. Ex: Concentrated or strong tea Less solute is dissolved than saturated. It can dissolve more. Ex: Diluted or weak tea Saturated & Unsaturated
Factors that affect solubility 1. Temperature: • For solids- ↑ temperature, ↑ solubility • For gases- ↑ temperature, ↓ solubility 2. Pressure: • For gases in liquid- ↑ pressure, ↑ solubilty
Super saturated solution- a solution that contains more solute than saturated solutions at the same temp/pressure
http://www.youtube.com/watch?v=D1PDE5OawuI&edufilter=iaYy5cltW5JHB2qLmddDPghttp://www.youtube.com/watch?v=D1PDE5OawuI&edufilter=iaYy5cltW5JHB2qLmddDPg
Henry’s Law- at a given temperature, solubility of a gas in a liquid is directly proportional to the pressure of a gas above the liquid. • Example: a soda
15.2 Solution Concentration • How much solute is dissolved in a specific amount of solvent is concentration
Describing concentration • Qualitative: • Concentrated or diluted
Describing concentration • Quantitative: • Percent by volume • Molarity (M) • Molality • Normality
Molarity: (M)(mol/L)= moles of solute liters of solution
Example 1 • A 100.5 mL intravenous solution contains 5.10 g of glucose (C6H12O6). What is the molarity of this solution? • Hint: glucose molar mass is 180. g/mol. • Practice 1-3
15-3 Colligative Properties of Solutions • Physical properties that are affected by the number of particles and not by their identity are colligative properties.
Examples of colligative properties include: • 1. vapor pressure lower • 2. boiling point elevation • 3. freezing point depression • 4. osmotic pressure
Solutions: • Solutes will not settle out • Will not scatter light • Cannot filter
15-4 Heterogeneous Mixtures • Suspensions: • Settles out • Scatters light • Can be separated by filtering • Are large particles (>1000 nm) • Examples: muddy water, fine sand in water
Colloids: • Don’t settle out • Scatters light • Can’t be filtered • Medium to small sizes (<1000 nm) • Examples: milk
Brownian motion is the random movement of colloid particles. • Causes collision of particles preventing them from settling.
Tyndall effect: • Particles scatter light • Can be seen in suspensions & colloids • Examples: ray of light through fog or smoke
Acid & Bases Chapter 19 • Acids, bases, & salts are electrolytes because they conduct electrical current. • An indicator is a compound that changes color to indicate the presences of an acid or base.
Characteristics of acids • Sour taste • Reacts with metals • Neutralizes bases • affects indicators • turns blue litmus red/pink • keeps phenolphthalein colorless • turns methyl orange red
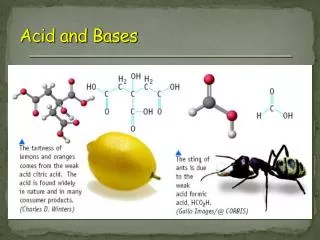
Common Acids: • Citric acid- in citrus fruits • (Lemons, oranges, etc) • Acetic acid- vinegar • Malic acid- apple • Butyric acid- sour butter • Lactic acid- sour milk; builds up in muscles during exercise.
Industrial acids: • Sulfuric acid- (H2SO4) most manufactored chemical in US; fertilizers • Phosphoric acid- (H3PO4) fertilizers; detergents • Nitric acid- (HNO3) fertilizers; explosives • Hydrochloric acid- (HCl) used to “pickle” steel (remove surface impurities)
Review: Naming acids • 1. Binary acids- (only 2 elements) “hydro________ic acid” • Ex. HBr • Ex. H2S • 2. Oxyacids- (has polyatomic ions) “________ic acid” • Ex. HClO3 • Ex. H2SO4
Characteristics of Bases • tastes bitter • feels slimy • affects indicators • pink litmus turns blues • phenolphthalein turns bright pink • methyl orange turns yellow
Common Bases: • Ammonia- (NH3) cleaning agent • Lye- (NaOH) draino • Milk of magnesia- (Mg(OH)2) laxative • Lime- (Ca(OH)2) mortar for bricks, foul lines on fields
What is pH? • pH is the negative logarithm of H+1 ion concentration • pH= -log[H+] • pH range- 014 • pH scale: acids 0-6.99, neutral 7, base 7.01-14
Example 2 • What is the pH of a solution with the concentration [H+]= 1.0 x 10-7 M? • Practice 4. What is the pH of [H+]=1.0x10-2? 5. What is the pH of [H+]=3.0x10-6? pOH? 6. What is the pOH of [OH-]=8.2x10-6?
Ex. 3 What is the concentration (Molarity) of H+ of an acidic solution with the pH of 3.20? • Practice: • 7. Calculate the concentration of H+ of an acidic solution with the pH of 6.55.
Neutralization • Neutralization Reaction- reaction between an acid & base to produce a salt & water • Salt- made of the positive ion (from base) & the negative ion (from acid) • Example: • Mg(OH)2 + HCl • Practice: • H3PO4 + Ca(OH)2 • Al(OH)3 + H2SO4
Acid is the sub. containing H Base is the sub. containing OH Ex. HCl & NaOH Acid is the sub. that loses the H+ Base is the sub. that gains the H+. involves conj. acid & bases H2O + NH4+ NH3 + H3O+ Arrhenius vs Bronted-Lowry
Practice: • Identify the following Bronsted-Lowry acids & bases. • HC2H3O2 +H2O H3O+ + C2H3O2-
Anhydrides are compounds that can become acids or bases when water is added. • Ex. Which is the anhydride? • CO2 + H2O H2CO3 • Practice. Which is the anhydride? • CaO + H2O Ca(OH)2 • H2O + SO2 H2SO4