Reaction Mechanisms
Reaction Mechanisms. Presented by Leticia Bonita Prince Newcastle University 4 th Year MChem Student. Aims of Today. Increase your understanding of reaction mechanisms and the bromination of alkenes. Increase your spatial skills and awareness using software designed by the CCDC.

Reaction Mechanisms
E N D
Presentation Transcript
Reaction Mechanisms Presented by Leticia Bonita Prince Newcastle University 4th Year MChem Student
Aims of Today • Increase your understanding of reaction mechanisms and the bromination of alkenes • Increase your spatial skills and awareness using software designed by the CCDC
What are Spatial Skills? • The ability to mentally manipulate 2-dimensional and 3-dimensional figures. • Important in chemistry, biology, medicine, mathematics, physics and everyday life.
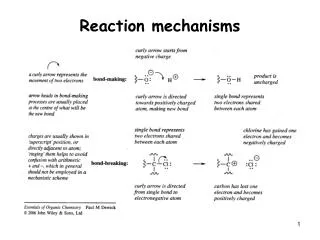
Curly Arrows • In a chemical reaction NEW BONDS ARE MADE and BONDS ARE BROKEN. A reaction mechanism illustrates how this happens. • Curly Arrows are used to highlight the MOVEMENT OF ELECTRONS. H+ + Cl- 2Cl. A HALF ARROW REPRESENTS 1 ELECTRON A FULL ARROW REPRESENTS 2 ELECTRONS Heterolytic Fission Homolytic Fission
Direction of Movement NUCLEOPHILE ELECTROPHILE
Nucleophiles • Electron Pair DONORS • Lewis Bases -CN, -OH, Cl-
Electrophiles • Electron Pair ACCEPTOR • Lewis Acids H+, CH3+, NO2+
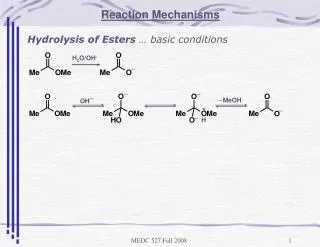
Electrophilic Addition to Ethene (Bromination) • Ethene has a π bond which has high electron density – NUCLEOPHILE • Bromine molecule will have a δ+ from Van der Waals’ - ELECTROPHILE
The WebCSD • Type in http://webcsd.ccdc.cam.ac.uk/teaching_database_demo.php • Answer the questions by typing the refcodes into the “find entry” box
THANK YOU! ENJOY THE REST OF YOUR DAY!