Balancing Equations Practice
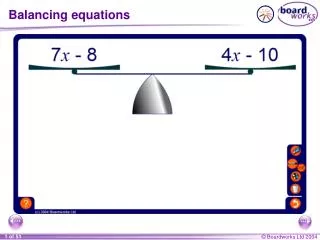
Balancing Equations Practice. ___ P + ___ O 2 ___ P 4 O 10. ___CO 2 + ___NH 3 ___OC(NH 2 ) 2 + __H 2 O. ___ Al 2 O 3 ___ Al + ___ O 2. __AlBr 3 + __K 2 SO 4 __KBr + __Al 2 (SO 4 ) 3. ___ Zn + ___ HCl ___ ZnCl 2 + ___ H 2. ___ HgO ___ Hg + ___ O 2.


Balancing Equations Practice
E N D
Presentation Transcript
The reaction below shows Chlorine (Cl) reacting with oxygen (O2) to form Cholrine oxide (ClO2). The mass of Chlorine is 25 grams and the mass of oxygen is 42 grams. What mass of Chlorine oxide is produced? a. 17 grams b. 39 grams c. 67 grams d. 71 grams
In the chemical reaction below, sodium (Na) and chlorine (Cl) combine to form sodium chloride (NaCl). 2Na + Cl2 → 2NaCl If the combined mass of the starting reactants is 50 grams, then the mass of sodium chloride formed once the reaction has reached completion • will be less than 50 grams. • will be exactly 100 grams. • will be exactly 50 grams. • will be between 50 and 100 grams
In a chemical reaction, the total mass of the reactants is 10.0 grams. After the reaction, the total mass of the products must be: • less than 10.0 grams. • exactly 20.0 grams. • more than 10.0 grams. • exactly 10.0 grams.