The Structure of Matter
320 likes | 544 Vues
The Structure of Matter. Physical Science Chapter 6. Review. Compound: atoms of two or more elements that are chemically combined Most of the matter around us is a compound or a mixture of compounds Compounds have properties unlike those of their elements

The Structure of Matter
E N D
Presentation Transcript
The Structure of Matter Physical Science Chapter 6
Review • Compound: atoms of two or more elements that are chemically combined • Most of the matter around us is a compound or a mixture of compounds • Compounds have properties unlike those of their elements • During a chemical change, a new substance is produced.
Chemical bonds • Forces that hold together the atoms in a compound • When atoms gain, lose, or share electrons they are forming chemical bonds.
Chemical Structure • The way the atoms are bonded in a compound
Chemical formulas • Used as shorthand for writing compounds. • NaCl is sodium chloride • Subscript – means written below • Tells us how many atoms of an element are in a compound • If there is no subscript, then there is one. • Example: H2O has 2 atoms of hydrogen and one atom of oxygen • The ratio of hydrogen atoms to oxygen atoms is 2 to 1
Chemical structure representations • Chemical formula – show how many of each type of atom there is • Water: H2O Methane CH4 • Structural formula – shows how atoms are arranged.
Chemical structure representations • Space filling model – shows relative volumes of the electron clouds. • Ball-and-stick model – shows bond angles
Effects of chemical bonds • Compounds with strong chemical bonds • Are rigid and difficult to break • Have high melting and boiling points • Compounds made of molecules • Have strong bonds within each molecule • Have weak attractions between molecules • Molecules are easy to separate • Lower melting and boiling points
Attractions between molecules • Some molecules have stronger attractions between them • Example: water • Has hydrogen bonding between molecules • Why water has a relatively high boiling point for a molecular compound
Discuss • Classify the following as mixtures or compounds • Air • CO • SnF2 • Pure water • Draw a ball-and stick model of a boron trifluoride, BF3, molecule. A boron atom is attached to three fluorine atoms. Each bond angle is 120 degrees and each bond is the same length. • Predict which molecules have a greater attraction for each other: C3H8O molecules in liquid rubbing alcohol or CH4 molecules in methane gas.
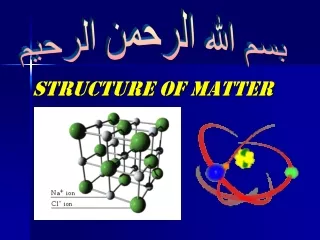
Ionic compounds • Ionic compound – a compound made up of two or more ions • Form networks of ions, not individual units. • Ionic bond – the force that holds the ions in an ionic compound together. • Ionic compounds have a net charge of zero, so the compound is electrically neutral.
Examples • NaCl • MgF2
Ionic compounds • Smallest unit is a formula unit. • Generally have high melting points and high boiling points. • Are usually crystalline solids at room temperature.
Covalent compounds • Covalent compounds are composed of molecules that are created when atoms share electrons • Covalent bonds – the bonds between atoms in a molecule. • Molecules are also neutral.
Examples • HCl • Cl2 • N2 • O2
Covalent compounds • Smallest unit is a molecule. • Generally have low melting points and boiling points. • Are usually liquid or gaseous at room temperature, but not always.
Discuss • Determine if the following compounds are likely to have ionic or covalent bonds. • Magnesium oxide, MgO • Strontium Chloride, SrCl2 • Ozone, O3 • Methanol, CH3OH • Identify which two of the following substances will conduct electric current, and explain why. • Aluminum foil • Sugar, C12H22O11 dissolved in water • Potassium hydroxide, KOH, dissolved in water
Polar molecules • Atoms in molecules don’t always share their electrons equally.
Examples • HCl • H2O
Polar molecule • Has a positive end and a negative end. • Example: stream of water
Nonpolar molecules • Do not have negative and positive ends. • Example: CO2 • Nonpolar vs. polar
Metallic Bonds • Occur between metal atoms • Atoms are closely packed together • Electron clouds overlap • Electrons move freely between atoms
Polyatomic ions in compounds • Poly means many • Polyatomic ions have more than one atom in them. • See Figure 10 on page 190
Compounds with polyatomic ions • They form compounds just like monatomic (one atom) ions do. • Examples • LiOH, lithium hyrdoxide • Mg(NO3)2, magnesium nitrate
Discuss • Compare bonds • Compare bonds • What is the difference between polar molecules and nonpolar molecules? • What are polyatomic ions? • Identify which of the bonds in calcium hydroxide, Ca(OH)2 are ionic and which are covalent.
Naming Ionic Compounds • List the (positive) cation first • Name is usually the same as the element • List the (negative) anion second • Change ending to –ide • See figure 2 on page 192
Examples • CaF2 • Calcium fluoride • Li2O • Lithium oxide • K2S • Potassium sulfide
Writing formulas for ionic compounds • The charge on the compound must add up to zero. • Add subscripts as needed
Examples • Cesium Oxide • Cs2O • Beryllium chloride • BeCl2 • Calcium Phosphide • Ca3P2
Transition Metals • Groups 3 – 12 • Can have more than one charge when forming compounds • Copper and oxygen can make CuO or Cu2O3 • To name them, we need to specify the charge of the cation using a roman numeral • CuO is copper (II) oxide • Cu2O3 is copper (III) oxide
Examples • Titanium (III) nitride • TiN • Fe2O3 • Iron (III) oxide • Iron (II) oxide • FeO