Specific Heat and Heat Capacity Units
60 likes | 306 Vues
Learn how to determine specific heat from given heat and temperature change data. Explore how different units and formulas are utilized in calculating heat capacities.

Specific Heat and Heat Capacity Units
E N D
Presentation Transcript
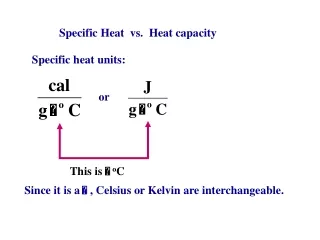
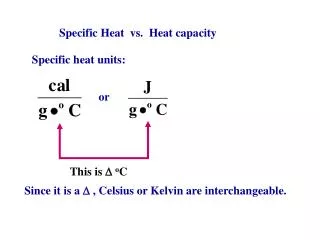
Specific Heat vs. Heat capacity Specific heat units: or This is oC Since it is a , Celsius or Kelvin are interchangeable.
Specific Heat vs. Heat capacity Specific heat units: or Heat Capacity units:
It took 74.9 J of heat to raise the temperature of 10.0 g of a substance 8.50oC. Determine the specific heat of the substance. Hint: focus on the desired units.
It took 74.9 J of heat to raise the temperature of a 10.0 g of a substance 8.50oC. Determine the specific heat of the substance. 74.9 J 0.881 = 10.0 g 8.50 oC The units match!
How many calories of heat are evolved if 44.7 g of Al undergoes a cooling change of 15.0 oC? (specific heat of Al = 0.214 cal/g•K) 44.7 g 15.0 K = cal K vs. oC? ’s are the same! WHY?