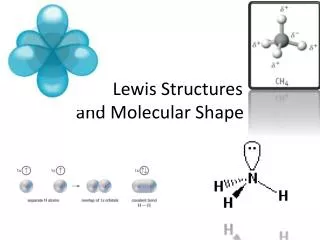
Molecular Shape
E N D
Presentation Transcript
Things to remember shape of molecule influenced by location of nuclei • nuclei go to certain locations because of its electron pairs
Use the Lewis Structure • Lewis structure is 2-D, but can help figure out 3-D shape
Atom Lewis Structure # Unpaired Electrons Bonding Capacity . H H 1 1 .. F, Cl, Br, I ·F: 1 1 .. . C, Si ·C· 4 4 . .. N, P ·N· 3 3 . O, S ·O: 2 2 · ·· Ne, Ar, Kr :Ne: 0 0 ·· Bonding Capacity ..
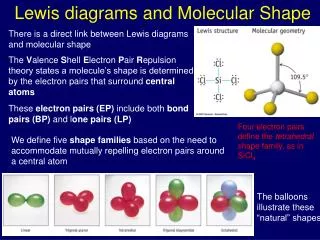
Molecular Shape • Determined by overlap of orbitals • Shape determined by two factors: • total # atoms & • # e- pairsin different locationson central atom • classify e- pairs as bonding or non-bonding
Molecular Shape & VSEPR • electron pairs repel each other: • want to be as far apart from each other as can be • non-bonding pairs take up a little more room than bonding pairs
Total # of electron pairs Orientation 1 or 2 linear: 2 electron pairs 180 apart 3 planar Triangular: 3 electron pairs 120 apart 4 tetrahedral: 3-D, 4 e- pairs 109 apart Orientation of Electron Pairs
2-Atom Molecules • atoms located next to each other • linear molecules!
Linear Bent 3-Atom Molecules • linear or bent
.. .. .. .. CO2 Lewis structure: O :: C :: O 4 bonding pairs but only2 regionsof electron density: 180 apart so CO2 is linear
Lewis Structure of H2O: H:O: H .. Count up the electron pairs: H2O .. • 2 bonding pairs between each O and H • 2 nonbonding pairs on O • 4 different regions of electron density
H2O • 4 electron pairs are 109 apart • nonbonding pairs spread out a bit more • squeeze bonding pairs together so bent shape H – O H
3-Atom Molecules • Triangular? Yes, ozone (O3) is triangular
4-Atom Molecules • Two possibilities: • Trigonal Planar – in 1 plane • Trigonal Pyramidal
4-Atom Molecules: Trigonal Planar Bond angles =120 All 4 atoms lie in the same plane aren’t likely to see this because thesemolecules don’t obey octet rule!
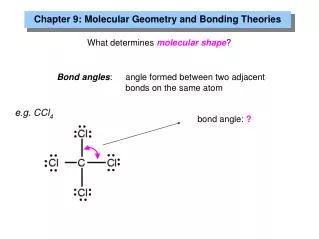
.. .. F .. .. .. .. .. F .. .. BF3: Trigonal Planar • B has 3 valence electrons • very small atom e • each F has 7 valence electrons • total = 3 X 7 + 3 = 24 e- : F : B These molecules don’t obey octet rule - so aren’t likely to see them - 3 regions - FBF = 120 ..
4-Atom Molecules: AX3 shape most likely to see
.. Lewis Structure: H : N : H .. NH3 H • 3 bonding pairs • 1 non-bonding pair • 4 regions • little less than 109 apart • shape: trigonal pyramidal
5 Atoms & Tetrahedrons Tetrahedral means 4 faces: - 1 atom in center - 4 atoms at the points
H Lewis structure = H : C : H .. H CH4 .. • 4 bonding pairs • 4 regions of electron density • electron pairs are 109 apart
Summary of Molecular Shapes • start with Lewis Structure! • look at # regions of e-density on central atom • look at # atoms bonded to central atom
Molecular Polarity • Look at shape of molecule: • non-polar molecular is symmetrical • polar molecular is asymmetrical
Molecular Polarity • molecule is symmetric if: • electrical charge on 1 side is same as electrical charge at matching point on opposite side • “pull” of one polar bond is offset by “pull” of another polar bond
Nonpolar Symmetrical Molecules are Non-polar
Asymmetric Molecules are Polar