Bonding
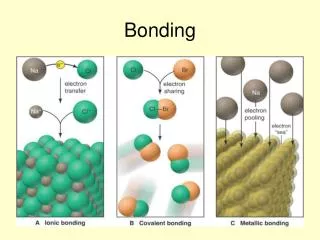
Bonding. Types of Bonds. Ionic Covalent Metallic. Metallic Bonds. Electrons are shared by many atoms Electrons free to move Two or more metals. Metallic Compounds. Generally high MP Hard & lusterous Less brittle Conductors. Metallic Bonds. No debate about metallic bonds

Bonding
E N D
Presentation Transcript
Types of Bonds • Ionic • Covalent • Metallic
Metallic Bonds • Electrons are shared by many atoms • Electrons free to move • Two or more metals
Metallic Compounds • Generally high MP • Hard & lusterous • Less brittle • Conductors
Metallic Bonds • No debate about metallic bonds • Easy to identify
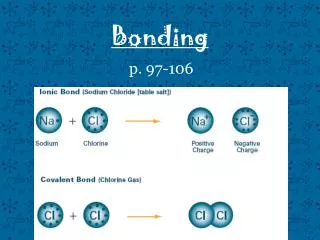
Ionic Bonds • Electrons are transferred from one atom to another creating (+) & (-) ions • Metal & nonmetal
Ionic Compounds • Held together by electrostatic charge • Very high MP • Brittle
Covalent Bonds • Electrons are shared by two atoms • Two nonmetals • Weaker than ionic
Covalent Compounds • Molecules • Low MP • Two nonmetals • Flexible
Molecule • Any compound that can exist as an entity by itself
Distinguishing Bonds • Distinguishing ionic & covalent bonds can be difficult, but generally determined by differenceelectronegativity
Bonds Types • Ionic • Polar covalent • Non polar covalent
Bond Types • Ionic: DEN > 1.5-1.8 • Covalent: DEN < 1.5-1.8 • Polar Covalent: 0.5<DEN<1.5 • Non polar covalent :DEN< 0.5 • Not absolute
Coordinate Covalent Bonds • A covalent bond in which the two electrons are donated by one atom
Classify Bonds • Na-F H-Cl • C-O Mg-O • Fe-Fe
Dipole • Polar bonds • Polar molecules
Dipole H F d+ d-
Ionic Bonding - +
Covalent Bonding Occurs when electron orbitals overlap
Orbitals On the board Max 2 e- per orbital
Hybridization • When s, p, and/or d orbitals (electron clouds) mix to make a new type of multi-lobed orbital
Hybrid Orbitals • sp dsp3 • sp2 d2sp3 • sp3
Electron Cloud Repulsion • In molecules each electron cloud repels other clouds enough to spread as far apart as possible
VSEPR • Valance Shell Electron Pair Repulsion • Electron pairs repel each other to spread out as much as possible
Bonding OrbitalsType HybridVSEPR • 2 lobes sp AX2 • 3 lobes sp2 AX3 • 4 lobes sp3 AX4 • 5 lobes dsp3 AX5 • 6 lobes d2sp3 AX6
Bonding Orbital Shape • 2 lobes: Linear 180o • 3 lobes: Trigonal planar 120o • 4 lobes: Tetrahedral 109.5o • 5 lobes: Hexahedral 120&180o • 6 lobes: Octahedral 90&180o
Bonding Electron Dot Diagrams • Electron dot diagrams that go through 4 singles before any electrons are paired up
1A: 1 single 2A: 2 singles3A: 3 singles4A: 4 singles 5A: 1 pair & 3 singles6A: 2 pair & 2 singles7A: 3 pair & 1 single8A: 4 pair
Lewis Dot Diagrams • Representation of valence electrons and bonds in a molecule or polyatomic ion
Drawing LDDs Draw the bonding electron dot diagram for each element in the molecule with the element with the most unpaired e- near the center
Drawing LDDs • If there is more than one carbon, connect the carbons by connecting single dots between one carbon & another
Drawing LDDs • Connect a single dot on one atom to a single dot on another (never two on the same atom)(never connect one dot to more than one other dot)
Drawing LDDs • Repeat connecting the dots until all singled dots are connected making sure to obey the octet rule if possible
Drawing LDDs • Recognize polyatomic ions • H2CO3: CO3-2 is a polyatomic ion; thus, the three Os must connect to the C
Drawing LDDs • Redraw the molecule neatly making sure to include all dots
Draw LDDs for: • BeCl2 H2O • BF3 C2H6 • CH4 C3H6 • NH3 CH2O
Draw LDDs for: • SO2 H2O • H2CO3 SO4-2 • CO2 CO3-2 • HCN C2H6O
Drill: Draw LDDs for: • PH3 PO3-3 • HCN C4H8O
Check HW • Problem 1 • Page 202
Draw LDDs for: • PO4-3 P2O7-4 • K2SO4 C5H8O