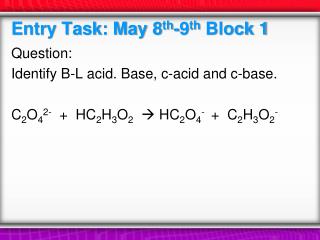
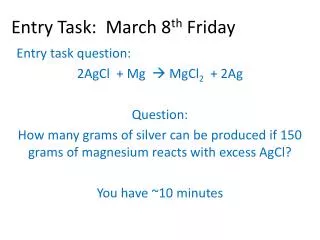Entry Task: Nov 21 st Block 2
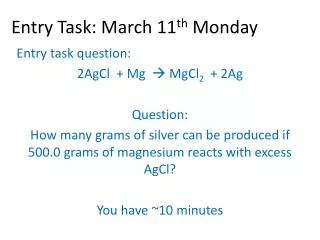
Entry Task: Nov 21 st Block 2. Question: What is the molality of a 200 g solution with 32 grams of NaCl ? TURN IN ENTRY TASK SHEETS!! You have 5 minutes!. Agenda:. Discuss Ch. 13 sec. 4 pHet Concentration and Molarity Lab HW: Solutions ws. I can ….

Entry Task: Nov 21 st Block 2
E N D
Presentation Transcript
Entry Task: Nov 21st Block 2 Question: What is the molality of a 200 g solution with 32 grams of NaCl? TURN IN ENTRY TASK SHEETS!! You have 5 minutes!
Agenda: • Discuss Ch. 13 sec. 4 • pHet Concentration and Molarity Lab • HW: Solutions ws
I can … • Express a concentration of a solution in different ways- %, ppm, mole fraction, Molarity, & Molality
mass of A in solution total mass of solution Mass Percentage Mass % of A = 100
mass of A in solution total mass of solution mass of A in solution total mass of solution Parts per Million andParts per Billion Parts per Million (ppm) ppm = 106 Parts per Billion (ppb) 109 ppb =
13.3 problem (a) Calculate the mass percentage of NaCl in a solution containing 1.50 g of NaCl in 50.0 g of water. 1.50 51.5g solution X 100 = 2.91%
13.3 problem (b) A commercial bleaching solution contains 3.62 mass % sodium hypochlorite, NaOCl. What is the mass of NaOCl in a bottle containing 2.50 kg of bleaching solution? X 2500g 36.2 100 100 X = 90500 X = 90.5g
moles of A total moles in solution XA = Mole Fraction (X) • In some applications, one needs the mole fraction of solvent, not solute—make sure you find the quantity you need!
mol of solute L of solution M = Molarity (M) • You will recall this concentration measure from Chapter 4. • Because volume is temperature dependent, molarity can change with temperature.
mol of solute kg of solvent m = Molality (m) Because both moles and mass do not change with temperature, molality (unlike molarity) is not temperature dependent.
Changing Molarity to Molality If we know the density of the solution, we can calculate the molality from the molarity, and vice versa.
13.4 problem • What is the molality of a solution made by dissolving 36.5 g of naphthalene (C10H8) in 425 g of toluene (C7H8)? 1 mole 35g = 0.28 mole = 0.670 kg 0.425 kg 128.1 g
13.5 problem • A commercial bleach solution contains 3.62 mass % NaOCl in water. Calculate (a) the mole fraction and (b) the molality of NaOCl in the solution. 3.62 % in water means 3.62 g in water 3.62 g – 100 = 96.38 g of water 3.62g 1 mole = 0.0489 mole 0.0489 mole 5.398 mole 74 g 9.06 x 10-3 96.38g 1 mole = 5.35 mole 18 g 0.0489 mole + 5.35 mole = 5.398 mole
13.5 problem • A commercial bleach solution contains 3.62 mass % NaOCl in water. Calculate (a) the mole fraction and (b) the molality of NaOCl in the solution. 3.62 % in water means 3.62 g in water 3.62 g – 100 = 96.38 g of water 3.62g 1 mole = 0.0489 mole 74 g 0.0489 mole = 0.507 m 0.09638 kg
13.21 13.21 a) Calculate the mass % of Na2SO4 in solution containing 14.7 g Na2SO4 in 345g H2O. b) Ore containing 7.35 g of silver per ton of ore. What is the concentration of silver in ppm? 14.7 g X 100 = 4.09% 14.7 + 345 g 7.35 g 1.10 x 10-6 ton = 8.09 ppm 1 g
13.23 13.23 Calculate the mole fraction of methanol, CH3OH, in the following solutions: a) 7.5 g CH3OH in 245 gH2O; b)55.7 g CH3OH in 164g CCl4. 7.5 g 1 mole = 0.23 mole of methanol 32.01 g 245 g 1 mole = 13.6 mole of water 18.0 g 0.23 mole of methanol = 0.017 mole fraction 13.6 + 0.23 mole solution
13.23 13.23 Calculate the mole fraction of methanol, CH3OH, in the following solutions: a) 7.5 g CH3OH in 245 gH2O; b)55.7 g CH3OH in 164g CCl4. 55.7 g 1 mole = 1.74 mole of methanol 32.01 g 164 g 1 mole = 1.07 mole of CCl4 153.81 g 1.74 mole of methanol = 0.620 mole fraction 1.74 + 1.07 mole solution
13.25 13.25 Calculate the molarity of the following aqueous solutions: a) 10.5g KCl in 250.0 ml of solution; b) 30.7g LiClO4•3H2O in 125ml o solution; c) 25.0 ml 1.50M HNO3 diluted to 0.500L 10.5 g 1 mole = 0.14 mole = 0.560 M 74 g 0.250 L 30.7 g 1 mole = 0.191 mole = 1.53 M 106.397 g + 3(18g) 0.125 L (0.025L) (1.50) = (x)(0.500L) = 0.075 M
13.27 13.27 Calculate the molality of each the following solutions; a) 13.0 g benzene, C6H6, dissolved in 17.0 g of CCl4; b) 4.75 g NaCl dissolved in 0.250 L of water whose density is 1.00g/ml. 13.5 g 1 mole = 0.167 mole = 9.79 m 78 g 0.017 kg 4.75 g 1 mole = 0.081 mole = 0.325 m 58.45 0.250 kg
13.29 13.29 A sulfuric acid solution containing 571.6g of H2SO4 per liter of solution has a density of 1.329 g/cm3 , Calculate a) the mass percentage; b) the mole fraction; c) molality; d) molarity of H2SO4 571.6 g X 100 = 43.0% 1329 g 571.6 g 1 mole = 5.83 moles of H2SO4 98 g 757.4 g 1 mole = 42.1 moles of H2O 18 g 5.83 moles of H2SO4 = 0.122 5.83 + 42.1 = 47.93 moles
13.29 13.29 A sulfuric acid solution containing 571.6g of H2SO4 per liter of solution has a density of 1.329 g/cm3 , Calculate a) the mass percentage; b) the mole fraction; c) molality; d) molarity of H2SO4 5.83 mol = 7.70 m 0.7574 kg 5.83 moles of H2SO4 = 5.83M 1 L
13.30 13.30 A solution containing 80.5 g of ascorbic acid, C9H8O6, dissolved in 210 g of water has a density of 1.22 g/cm3 , Calculate a) the mass percentage; b) the mole fraction; c) molality; d) molarity of C9H8O6 80.5g X 100 = 27.7% 210 g + 80.5g 80.5 g 1 mole = 0.457 moles of C9H8O6 176.1 g 210 g 1 mole = 11.7 moles of H2O 18 g 0.457 moles of C9H8O6 = 0.0376 0.457+ 11.7 = 12.16 moles
13.30 13.30 A solution containing 80.5 g of ascorbic acid, C9H8O6, dissolved in 210 g of water has a density of 1.22 g/cm3 , Calculate a) the mass percentage; b) the mole fraction; c) molality; d) molarity of C9H8O6 0.457 mol = 2.18 m 0.210 kg 1 L 290.5 g 1 mL = 0.238L 1.22 g 1000 mL 0.457 moles of C9H8O6 = 1.92 M 0.238 L