Understanding Kinetics: Key Factors Influencing Reaction Rates
This video outlines the essential elements required for a chemical reaction, emphasizing the significance of activation energy and effective collisions. It explains collision theory, noting that not all collisions result in reactions—only effective ones do. The rate of reaction is determined by factors such as the nature of reactants, temperature, concentration, pressure, surface area, and the presence of catalysts. By exploring the dynamics of molecular interactions, this video provides insights into how fast reactions can occur and how various factors can alter these rates.

Understanding Kinetics: Key Factors Influencing Reaction Rates
E N D
Presentation Transcript
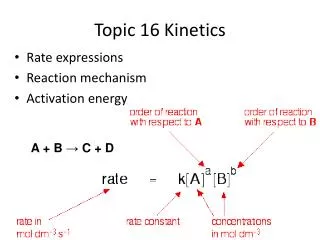
2 things needed for a reaction to occur • X amount of activation energy • Effective collisions Particle Diagram of Collision NO + O3 NO2 + O2 Activated Complex is NOT in equation!
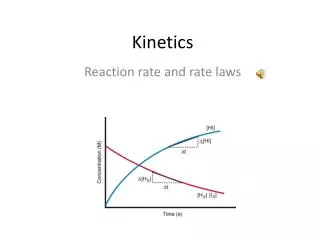
Kinetics tells how fast a reaction will occur • Reaction rate = speed of the reaction • Depends on Frequency of collisions (how often they occur) and efficiency of collisions • Rate = Concentration of reactant or product time Appearance of products Disappearance of reactants
Collision Theory • Molecules must collide in order to react • Effective collisions lead to formation of products • Effective Collisions = • Energetic • Favorable orientation • Ineffective collisions do not lead to products
Why Do Collisions Have to be Energetic? • KE of reactants is used to overcome reaction barrier • KE is transformed into PE
Factors that determine reaction rates • Nature of reactants (ions vs molecules)
Nature of the reactants:Ions or Molecules? • Ions in solution react quickly • Covalently bonded molecules react slowly • takes time to break all those bonds! • 2 gas phase reactants react more quickly than 2 liquids or 2 solids
Factors that determine reaction rates • Nature of reactants (ions vs molecules) • Temperature
Increase in Temperature • Temp = measure of average KE of molecules in system • faster molecules are moving, will collide more often • faster molecules are moving, more energetic the collisions
Factors that determine reaction rates • Nature of reactants (ions vs molecules) • Temperature • Concentration
Concentration • Increase in concentration: • more particles per unit volume – more collisions in given amount time
Factors that determine reaction rates • Nature of reactants (ions vs molecules) • Temperature • Concentration • Pressure (gases only)
Pressure • systems involving gases changing pressure analogous to changing concentration • Pressure, # particles per unit volume • Increase reaction rate • ↓ Pressure, ↓ # particles per unit volume • Decrease reaction rate
Factors that determine reaction rates • Nature of reactants (ions vs molecules) • Temperature • Concentration • Pressure (gases only) • Surface Area
Surface Area • Higher surface area • more particles exposed for reaction • Higher surface area means smaller particle size • (For heterogeneous reactions)
Homogeneous Reaction: • all reactants in same phase • Heterogeneous Reaction: • reactants in different phases
Factors that determine reaction rates • Nature of reactants (ions vs molecules) • Temperature • Concentration • Pressure (gases only) • Surface Area • Presence of a catalyst
Catalyst • Substance that increases rate of reaction without itself being consumed • does not participate in reaction • Lowers the activation energy for the reaction