Chapter 1 Soil Mineral Material
Chapter 1 Soil Mineral Material. Section 1 The chemical and mineral constitutes of soil parent minerals 一 、 The chemical constitutes of soil parent minerals. Soil. Mineral Material 38% , (95%). Organic matter 12% , (5%). Solution (15-35%). Air (15-35%). Distributive law.

Chapter 1 Soil Mineral Material
E N D
Presentation Transcript
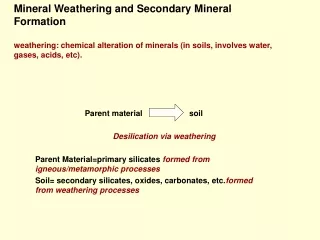
Section 1 The chemical and mineral constitutes of soil parent minerals 一、The chemical constitutes of soil parent minerals Soil
Mineral Material 38%,(95%) Organic matter 12%,(5%) Solution (15-35%) Air (15-35%)
Distributive law (1)The oxygen( O) and silicon( Si) are main two elements in the earth crust. The second elements are iron and aluminum. Four elements occupy about 88.7% weight of the earth crust. (2)The essential elements for plant growing are very low and distribute unbalance in earth crust.
二、The mineral constitutes in the soil According to the crystallize appearance of the mineral, the mineral can be divided into: Crystallization mineral and no-crystallization mineral According to the source of the mineral, the mineral can be divided into: Primary mineral and secondary mineral
①The silicate and aluminosilicates are main compositions in the primarymineral. ② The feldspar and quartz are main mineral in the primarymineral. ③The primarymineral is an important source of the plant nutrient . 二、The mineral constitutes in the soil Primarymineral formed from cooling the molten(lava) rock.
Section 2 Clay Mineral Clay minerals are mostly newly formed crystals reformed from the soluble products of the primary minerals and may be considered secondary minerals. 一、Layer silicate clay mineral (一)Structure characteristics 1、Basic construction unit
(1) Basic Building Blocks for Clay Minerals • Silicon - Oxygen Tetrahedron(Si2O5-2)
Basic Building Blocks - 2 • Aluminum Octahedral (Gibsite Sheet) • Al(OH)6-3
Silicate Clay Minerals • Aluminosilicates have a definite crystalline structure. Formed by the alteration of existing minerals or by synthesis from elements. • < .002 mm in size (only seen with aid of electron microscope) • Two basic kinds of clay minerals 1 : 1 and 2 : 1
(2) Types of Clay Minerals • 1) Kaolinite a 1: 1 clay mineral • 1 silica sheet and 1 gibsite sheet • .0072 cm thick
2 : 1 CLAY MINERALS • 1. Hydrous Mica or Illite • replacement of Si4+ with Al3+ in 25% of tetrahedrals, each substitution leaves 1 neg charge site also K+ ions are a bridge between layers and hold them tightly together K Space
2. Montmorillonite • Mg++ replaces Al+++ in Octahedral layer
2:1:1 Clay minerals A layer of chlorite has 2 silica tetrahedra, an alumina octahedra, and a magnesium octahedra sheet. They are often called 2:2 or 2:1:1 type Clay minerals
4. Isomorphous Substitution • Isomorphous Substitution is the substitution of one ion for another of similar size but low positive valence. • E. g. Al3+ for Si4+ or Mg2+ for Al 3+
The regulation ofIsomorphous Substitution in soil: 1. Notice that most substitutions are by ions with lower charge (less positive) than the ones being replaced. 2. The total negative charge from the anions (the oxygens) remains unchanged,but lower positive charge because of substitution results in an excess negative charge at that general site.
Four groups: Kaolinit Montmorillonite Hydrous mica (illite) Chlorite (二)、Types and general characteristics of silicate clay minerals
Kaolinit group Properties: (1) 1:1 type (2)Almost no substitution of Al3+ for Si4+,or Mg2+ for Al3+ has occurred in kaolinite, so the net negative charge is low. (3)It does not shrink and swell. (4)The colloidal characteristics are weaker. Kaolinit group is in moist, warm to hot, subhumid and humid leached soils.
Montmorillonite group • Properties: • (1)2:1 type • (2) Large expansion • (3) Isomorphous substitution is common inMontmorillonite, which has most of charge. • (4)The colloidal characteristics are strong. • Montmorillonite group is in arid to humid soils having limited leaching.
Hydrous mica (illite) group • Properties: • (1) 2:1 type • (2) non-expanding • (3)Large potassium ions holding adjacent layers together • (4)Slight to moderate swelling • Hydrous mica (illite) groupis subhumid and cool areas, parent rock with micas.
Chlorite group Properties: (1)2:2 or 2:1:1 type Clay minerals (2)Without swell (3)Low cation exchange capacities This group previously formed in marine sediments.
二、Sesquioxide clays (metal oxides and hydrous oxides) These clays can be either amophous or crystalline. They do not swell, are mot sticky, and do not behave as do the silicate clays. (一)iron oxide (Fe2O3) Goethite(a-FeOOH) Hematite(a- Fe2O3) Iron oxide and iron hydrate commonly color the soils various shades of red to yellow, respectively.
(二) Aluminum hydroxide (Al(OH)3 or Al2O3·xH2O) (三) Allophane (xAl2O3·ySiO2·nH2O) (四) Silicon oxide
第三节 我国土壤粘土矿物分布规律 一、风化和成土作用与粘土矿物组成的关系 1、风化递变学说 白云母经风化很容易形成水化云母,随着风化和淋溶程度的发展,云母类型矿物可能依次顺着伊利石、蛭石、蒙脱石、高岭石以至三水铝石的方向递变。
老化 结晶态粘土矿物 非结晶态粘土矿物 酸胶基 + 碱胶基 Al2O3·nH2O Fe2o3·nH2O SiO2·nH2O 2、风化合成学说(自然合成说) 粘土矿物也可能不是直接从原来的矿物变过来的,而是由化学风化所分离出来的简单风化产物在一定条件下重新组合沉淀而成。即:
二、我国土壤粘土矿物分布规律 (一)水云母区 包括新疆、内蒙古高原西部、柴达木盆地、青藏高原大部。土壤粘土矿物以水云母为主,其次为蒙脱石和绿泥石。 (二)水云母-蒙脱石区 包括内蒙古高原东部、大小兴安岭、长白山地和东北平原大部分。土壤粘粒中蒙脱石明显增多。 (三)水云母-蛭石区 包括青藏高原东南边缘山地、黄土高原和华北平原。西部山地土壤粘粒中绿泥石,东部多蛭石,华北平原土壤粘粒中蒙脱石也不少。 (四)水云母-蛭石-高岭区 包括秦岭山地和长江中下游平原,为一狭长的过渡地带,在适宜条件下,水云母、蛭石和高岭石都可成为土壤粘粒中的主要成分。
二、我国土壤粘土矿物分布规律 (五)蛭石-高岭区 包括四川盆地、云贵高原、喜马拉雅山东南端。土壤粘粒中云母退居次要成分,以蛭石和高岭为主。东部蛭石尤多,并多三水铝石;西部蛭石较少,氧化物含量很 高,山地土壤中水云母含量随海拔高度升高而增加。四川盆地土壤中还有不少蒙脱石。 (六)高岭-水云母区 包括浙、闽、湘、赣大部和粤、桂北部。土壤中粘粒部分结晶差的高岭石为主。东部不少水云母和蛭石伴存,铁铝氧化物含量也显著增多。 (七)高岭区 包括贵州南部、闽粤东南沿海、南海诸岛及台湾。
本章的主要内容是土壤矿物质的组成及粘土矿物的结构与性质。本章的主要内容是土壤矿物质的组成及粘土矿物的结构与性质。 真章的重点是粘土矿物的结构和性质,认真理解和掌握粘土矿物的结构和性质对学习好以后各章有重要的作用。可以帮助你正确理解土壤的许多理化性质。 主要掌握几种代表性的粘土矿物。 1、 2、 3、 本章小结
课堂速测 1. 砂质土全部由砂粒所组成,粘质土全部由粘粒所组成 ( )。 2. 在同一地区,土壤质地愈粘,则养分含量愈多( ) 3.南方红、黄壤中的粘土矿物以高岭石为主,而北方土壤 中以蒙脱石,伊利石为主( )。 4.母质是形成未来土壤的岩石、矿物的风化产物( )也是搬 运后形成的地面沉积体( ) 5. 不同土壤类型粘土矿物的组成不同( )。
课堂速测 • 6. 农业土壤土体的底土层就是指的母质层( )。 • 7. 任何土壤的矿物质组成中既含有原生矿物,也含有次生矿物( )。 • 8. 土壤矿物质的化学组成,一般N素含量很少( )。 • 9. 土壤中的粘粒均由次生矿物所组成并多以云母为主( )。 • 蒙脱石矿物晶架的顶,底层由离子键相连( );高岭石矿顶、底层由氧键相连( );伊利石矿物则由Si-O相连( )。 式中X表示补偿异价离子置换引起的电荷亏缺的层间可交换阳离子。