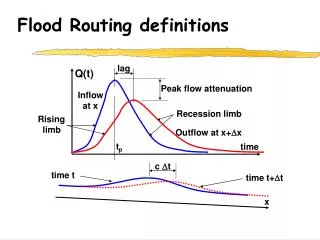
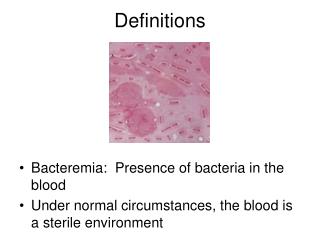
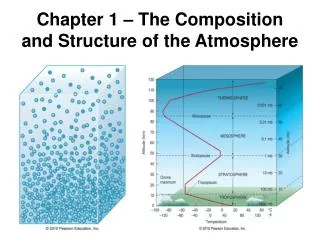
Isomerism – Some Definitions
Isomerism – Some Definitions. Stereochemistry - chemistry in 3-D - spatial relations of atoms in molecules stereoisomers - compounds with the same molecular formula, but which differ in the spatial arrangement of

Isomerism – Some Definitions
E N D
Presentation Transcript
Isomerism – Some Definitions Stereochemistry - chemistry in 3-D - spatial relations of atoms in molecules stereoisomers - compounds with the same molecular formula, but which differ in the spatial arrangement of their atoms. • An understanding of biochemistry requires an awareness of the stereochemistry of molecules.
Chirality • chiral - Greek for hand - used to describe objects that cannot be superimposed on their mirror images. • achiral - used to describe objects that can be superimposed on their mirror images. • achiral objects must have a plane of symmetry – an imaginary line dividing an object in half so that one half is the mirror image of the other
Chirality • chirality - a property of handedness which determines a molecule’s biological behavior. • For organic molecules, the chirality of the carbon atom is important. • Carbon forms 4 bonds. • Carbon bonds form a tetrahedron in 3-dimensional space. • A chiral carbon atom is one with 4 single bonds which are attached to 4 unlike atoms or groups of atoms. • A chiral carbon is also referred to as a stereocenter.
Determination Of Stereocenters To identify chiral carbons in a molecule, look for carbons which contain only single bonds and are attached to 4 unlike atoms or groups.
Stereoisomers • For a molecule with n stereocenters, the maximum number of stereoisomers possible is 2n
Plane of Symmetry • achiral objects must have a plane of symmetry – an imaginary line dividing an object in half so that one half is the mirror image of the other Chiral Achiral, plane of symmetry no chiral carbons
Enantiomers Enantiomers - a pair of stereoisomers that are nonsuperimposable mirror images of each other - enantiomers are chiral molecules - they are always in pairs.
Enantiomers • All physical and chemical properties of enantiomers are identical except: • 1. They rotate polarized light in opposite directions and • 2. They react with other chiral molecules at different rates (major biochemical consequences). • 3. Enantiomers have identical melting points, boiling points, densities, and rates of reaction with achiral molecules.
Optical Activity • Ordinary light: light waves vibrating in all planes perpendicular to its direction of propagation • Plane-polarized light: light waves vibrating only in parallel planes • Polarimeter: an instrument for measuring the ability of a compound to rotate the plane of plane-polarized light • Optically active: showing that a compound rotates the plane of plane-polarized light
Optical Activity Dextrorotatory: clockwise rotation of the plane of plane-polarized light (+) Levorotatory: counterclockwise rotation of the plane of plane-polarized light (-) • Two members of a pair of enantiomers rotate the plane of polarized light in opposite directions by the same amount of rotation. • A racemic mixture is one that contains equal concentrations of 2 enantiomers so optically inactive
The R,S System • The first step in assigning an R or S configuration to a stereocenter is to arrange the groups on the stereocenter in order of priority • priority is based on atomic number • the higher the atomic number, the higher the priority If priority cannot be assigned on the basis of the atoms directly attached to the stereo center look at the next atom and continue to the first point of difference.
The R,S System • To assign an R or S configuration 1.assign a priority from 1 (highest) to 4 (lowest) to each group bonded to the stereocenter 2.orient the molecule in space so that the group of lowest priority (4) is directed away from you; the three groups of higher priority (1-3) then project toward you 3.read the three groups projecting toward you in order from highest (1) to lowest (3) priority 4. if reading the groups 1-2-3 is clockwise, the configuration is R; if reading them is counterclockwise, the configuration is S
The R,S System -In determining R and S on a stereocenter You must look for the first point of difference. –CH2–OH 6 8 C to O –CH2–CH3 6 6 C to C –CONH2 6 8,8,7 C to O, O (double bond) then N –CHO 6 8,8,1 C to O, O (double bond) then H –CH2–H 6 1 C to H –OH 8 O
Fisher Projections • Fischer projections - a shorthand way to draw stereomolecules for non-artists (i.e. chemists and students) - they are used to determine if 2 molecules are identical or different and whether 2 formulas are mirror images. • a two dimensional representation for showing the configuration of tetrahedral stereocenters • horizontal lines represent bonds projecting forward • vertical lines represent bonds projecting to the rear
Fisher Projections • The virtue of Fischer projections is that they can be easily used for molecules with more than one chiral center. • Lets take 2, 3-dibromobutane as an example: This molecule is still not superimposible on the first one by sliding it, so it must be an enantiomer • A Fischer projection can never be removed from the plane of the paper. • 1800 rotation is allowed, 900 or 2700 rotations are not allowed.
Diastereomers • Diastereomers are chiral isomers but are not mirror images. (a) and (c), for example, are diastereomers
Diastereomers • Diastereomers – possess different physical properties, e.g. boiling point, melting point, solubility, etc