Ions in Solids and Solutions
240 likes | 266 Vues
Learn about ions in ionic solids, hydrated ions in solutions, bonding processes, precipitation reactions, and more in this comprehensive study. Explore examples and equations for better understanding.

Ions in Solids and Solutions
E N D
Presentation Transcript
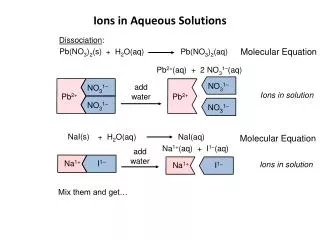
- - + - + + - - + Ions in ionic solids. Cl- Na+
δ- δ- δ- δ- δ- δ- δ- δ- δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ- δ+ Ions in solution. Hydrated ions - + Most electronegative Polar molecules O Oδ- H Hδ+ Hδ+ H
δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ- δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ δ+ Some bonds made Some bonds broken - - + - + + - - +
- Some bonds broken Some bonds made - + - + + - - +
+ Some bonds made - - + - + Some bonds broken - - +
Some bonds made Dissolving. Some bonds broken Exothermic Endothermic
For Mg(NO3)2, the ions present in solution are Mg2+(aq)+ 2NO3-(aq) Try Q1 from CI 5.1. Page 40 will help you.
Precipitation. A precipitate is a solid made when two solutions react.
Ag+ Ag+ NO3- NO3- NO3- Cl- Cl- Cl- Na+ Na+ Na+
NO3- NO3- Ag+ Na+ Na+ Cl- Ag+ Cl- Spectator ions
Some bonds made Some bonds broken Ag+ Cl- Ag+ Cl-
Some bonds made Precipitation. Some bonds broken Exothermic Endothermic
NO3- NO3- Ag+ Na+ Na+ Cl- Ag+ Cl- Spectator ions
NaCl(aq) + AgNO3(aq) AgCl(s) + NaNO3(aq) Spectator ions Na+(aq) +Cl-(aq) + Ag+(aq) + NO3-(aq) AgCl(s) + Na+(aq) + NO3-(aq) Ag+(aq) +Cl-(aq) AgCl(s) Ionic equation
HCl(aq) + NaOH(aq) H2O(l) + NaCl(aq) Spectator ions H+(aq) +Cl-(aq) + Na+(aq) + OH-(aq) H2O(l) + Na+(aq) + Cl-(aq) H+(aq) +OH-(aq) H2O(l) Ionic equation
More reactions of acids:Try Q5 from CI 5.1 A spectator ion is an aqueous ion on both sides of the equation!
Finally…. + 0.078nm 2+ 26
Water of crystallisation… Cu2+(aq) + SO42-(aq) + 5H2O(l) CuSO4.5H2O Try Q4 from CI 5.1
ICSD Chem Zone Lesson Dissolve & Dissociate_files The hyperlink below is a good flash animation of dissolving: http://www1.icsd.k12.ny.us/chemzone/lessons/03bonding/dissociate.htm