Unveiling the World of Elements: Atoms, Ions, and Compounds
200 likes | 237 Vues
Discover the fundamentals of chemistry with insights on elements, atoms, ions, and compounds. From Dalton's atomic theory to the modern understanding of isotopes and the periodic table, delve into the building blocks of matter. Explore isotopes, the structure of the atom, and important groups in the periodic table. Unravel the mysteries of ions, compounds, and allotropes of elements.

Unveiling the World of Elements: Atoms, Ions, and Compounds
E N D
Presentation Transcript
Chapter 4: Elements, Atoms, and Ions Chemistry 1020: Interpretive chemistry Andy Aspaas, Instructor
Elements • All substances on earth are made from a combination of 114 or so elements • 88 found in nature, others are man-made • Abundance: % found in nature • Oxygen most abundant on earth & in human body (by mass) • Abundances vary in different parts of environment • Each element has its own symbol • One or two letters - first is always capitalized
Dalton’s atomic theory • According to Dalton: • Elements are composed of tiny unbreakable particles called atoms • All atoms of a given element are identical • Atoms of a given element are different from those of any other element • Atoms of one element combine with atoms of other elements to form compounds • Atoms are indivisible by chemical processes
Conclusions from Dalton’s atomic theory • Law of constant composition • All samples of a pure compound contain the same proportions of the elements • Chemical formulas used to show those proportions • Law of conservation of mass • Atoms are never created or destroyed, only rearranged • Atoms of one element cannot change into atoms of another element • All atoms present at the beginning of a reaction are present at the end
Compounds and chemical formulas • A compound is a pure substance that is composed of atoms of two or more elements • Compounds are described by giving the number and type of each atom in the simples unit of the compound • Each element represented by letter symbol • Quantity of atoms of each element written as subscripts (subscript 1 never written) • Polyatomic groups in parentheses if more than one
Structure of the atom • J.J. Thomson investigated beams known as cathode rays • Made of tiny negatively charged particles called electrons • Smaller than a hydrogen atom! • Atoms of different elements produce the same electrons
Thomson’s plum pudding model • Atoms are indeed breakable, contrary to Dalton’s theory • Electrons are suspended in a positively charged electric field (to balance electrons’ neg. charge) • Mass of atom is due mostly to electrons • Atom is mostly empty space
Rutherford’s gold foil experiment • Tried to prove that atoms are mostly empty space • Shot “bullets” of alpha particles through thin sheet of gold atoms • Expected alpha particles to fire straight through • Most indeed did • But about 2% were deflected by very large angles • Disproved Thomson’s plum pudding model
Rutherford’s nuclear model • Most of atom’s mass is in a tiny dense center called the nucleus • Positively charged • Only 1/10 trillionth the volume of the atom • Nucleus’s positive charge balances electrons’ negative charge • Electrons fly around in the empty space surrounding nucleus
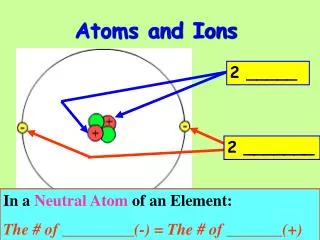
The modern atom • Nucleus composed of two types of particles • Protons: +1 charge • Neutron: 0 charge, mass similar to proton • Electrons: -1 charge, outside of nucleus
Isotopes • Number of protons defines which element an atom is • Called atomic number, found on periodic table • Atoms of an element with different numbers of neutrons are called isotopes • All isotopes of an element behave identically in chemical reactions • But have different masses • Identified by mass number • Mass number = # protons + # neutrons • Isotope symbols contain mass number, atomic number, and element symbol
Elements • Arranged in pattern called Periodic Table • Properties can be predicted based on position in table • Metals • About 75% of elements • Lustrous, malleable, ductile, conduct heat and electricity • Nonmetals • Dull, brittle, insulators • Metalloids or semi-metals • Properties of both metals and nonmetals
Groups and periods • Elements with generally similar chemical and physical properties are in the same column • Columns are called groups or families • Different numbering schemes • Rows are called periods
Regions of the periodic table • Main group = representative elements • “A” columns in label • Transition elements • All metals • Bottom rows: Inner transition elements or Rare earth elements • All metals • Inserted into periodic table after La and Ac
A few important groups • Group 8A: Noble gases • Colorless gases at room temperature • Non-reactive (inert) • Found in nature as single atoms uncombined • Group 7A: Halogens • Very reactive nonmetals • Exist as diatomic molecules in nature (Cl2, Br2, etc) • React with metals to form ionic compounds
Allotropes • Some solid nonmetallic elements can exist in different forms with different physical properties • Allotropes: these different forms • Different physical properties are from different arrangements of atoms in the solid • Allotropes of carbon: • Diamond • Graphite • Buckminsterfullerene (C60)
Ions • Ions are atoms or groups of atoms that carry an electrical charge • Cations: positive charge (loss of electrons) • Anions: negative charge (gain of electrons) • Unlike charges attract, so cations and anions are attracted to each other • Electrolytes: ions that dissolve in water and cause it to conduct electricity • Ionic compound: combination of cations and anions that form a pure substance with no total charge
Atomic structures of ions • Metals always form cations • Loss of one electron creates a +1 charge • Na = 11 e-, Na+ = 10 e- • Ca = 20 e-, Ca2+ = 18 e- • Cations have same name as uncharged metal • Charge can be determined from group number
Atomic structures of ions • Nonmetals form anions • Gain of 1 electron = -1 charge • Anions named by changing ending of element name to -ide • Fluorine: F + 1 e- = F- (fluoride ion) • Oxygen: O + 2 e- = O2- (oxide ion) • Charge determined by taking 8 - group number
Writing ionic formulas • Ionic compound must have no net charge • So change number of ions to cancel out positive and negative charges • Compound made of Mg2+ and Cl- • Must have formula MgCl2