Periodic Table
Learn how the periodic table is arranged and how to key it for different types of matter. Understand the properties and characteristics of elements in each group.

Periodic Table
E N D
Presentation Transcript
Aim 1 • To understand how the periodic table got its shape.
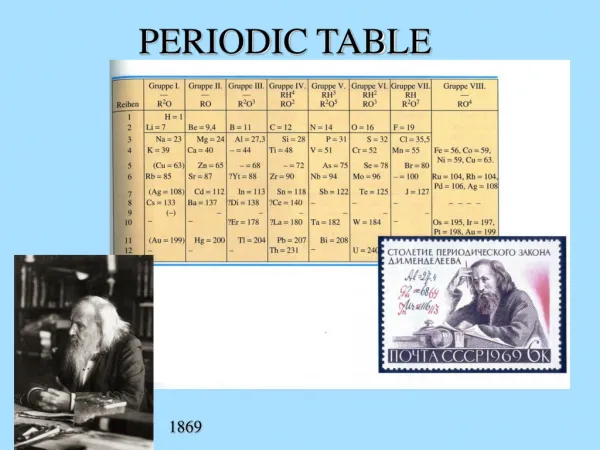
Mendeleev • Arranged the Periodic Table according to atomic mass • This resulted in gaps and the table being out of order!
Moseley • Arranged table by atomic number (or # of protons) • How the modern day periodic table is arranged • No more gaps! (we call this an observed regularity)
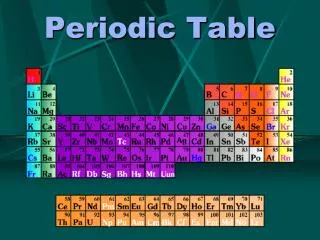
THE PERIODIC TABLE… • Is made up of Periods and Groups. • PERIODS = HORIZONTAL ROWS (run left to right) • Elements in the same period have the same number of energy levels/shells
Eight is great • OCTET = full VALENCE SHELL (8 e-) • ****except for Hydrogen (1) • Helium (2) • They only need 2 electrons to fill their valence shell
groups • GROUPS = VERTICAL COLUMNS (run up & down) • Elements in the same group have very similar chemical reactivity! This is based on valence electrons
Aim 2 • To “key” the periodic table in order to get as much information out of it as possible.
solids • Solids (s) – most elements are solids at STP • a. Definite VOLUME • b. Definite SHAPE
liquids • Liquids (l) – only TWO elements at STP (Br & Hg); • a. Definite VOLUME • b. Takes the SHAPE of the container
aqueous • Aqueous (aq) – a STATE of matter in which one substance (a solute) is dissolved in another (a solvent); a solution • ex: NaCl(aq) • Salt (NaCl) is the solute, Water is the solvent
gases • Gases (g) – H, N, O, F, Cl, & all of group 18 (noble gases) • a. No definite VOLUME • b. FILL their container uniformly
Diatomic elements • Elements that are UNSTABLE ALONE in nature • PAIR UP in order to fill their VALENCE SHELLS • Perfect sharing • Contains 2 identical atoms
Draw a lewis structure for 2 fluorine atoms • Put them together: • Use the 7- Up trick or:
Types of Matter: Solid and Liquid • 1. Metals: • make up MOST of table • LEFT of or BELOW “staircase”—except HYDROGEN • all SOLIDS (except Hg(l) • Must have ALL FOUR properties of a metal
Must have ALL FOUR properties of a metal: • 1. MALLEABLE : can be hammered/molded into sheets • 2. DUCTILE: can be drawn/pulled into wire • 3. have LUSTER: are shiny • 4. good CONDUCTORS: allow heat & electricity to flow through them
conductors • Metals are good conductors due to their “sea of mobile valence electrons” • Metals like to lose electrons to become positive ions
nonmetals • RIGHT of or ABOVE staircase • mostly GASES and SOLIDS @ STP—except Br(l) • NOT malleable/ductile; BRITTLE (shatter easily) • LACK luster (DULL) • NON or POOR conductors
Nonmetals • Nonmetals like to gain electrons to become negative ions
3. Metalloids (semi-metals) • Have TWO properties of a metal • ALONG staircase (between METALS & NONMETALS on • table)—except Al • There are 6 metalloids you have to know!
https://www.youtube.com/watch?v=hszrbXQKR-8 http://www.harcourtschool.com/activity/states_of_matter/
Group 1- ALKALI METALS (FAMILY) • Easily LOSE their one electron to become +1 ions • EXTREMELY reactive! Never found alone in nature • Contains the MOST reactive metal: FRANCIUM (Fr) • https://www.youtube.com/watch?v=uixxJtJPVXk • https://www.youtube.com/watch?v=m55kgyApYrY
What about hydrogen? • Not officially part of a group!! • Both a NONMETAL and a GAS • https://www.youtube.com/watch?v=v1TfPDlA1xE
Group 2: ALKALINE EARTH METALS (FAMILY) • LOSE their two electrons to become +2 ions • FAIRLY reactive! Never found alone in nature https://www.youtube.com/watch?v=DFQPnHkQlZM
Groups 3-12: TRANSITION METALS • Form COLORED IONS in solution. Group 12 is the HEAVY METALS and they don’t form colors. Tend to be UNPREDICTABLE!They can FORM DIFFERENT #’S OF BONDS
LEAST reactive group of metals • Nickel and Copper • https://www.youtube.com/watch?v=vwd4qsmUxwM
The Lanthanide/Actinide Series – two rows on bottom of table (detached) • – Elements 58 – 71 & 90 - 103 • Actually belong to the TRANSITION METALS
Groups 13-16: BCNO groups • MISCELLANEOUS groups • Metals, nonmetals, & metalloids found along the staircase (many different properties)
Group 17: HALOGENS (FAMILIY) • https://www.youtube.com/watch?v=J7b2aBKa6-U • Like to gain 1 electron to become ions with -1 charge • Form SALTS/ /COMPOUNDS • (a metal and a nonmetal) called HALIDES
Contains the most REACTIVE nonmetal: FLUORINE All NONMETALS making up the group Three states of matter found in group
Group 18: NOBLE GASES (FAMILY) • UNREACTIVE or INERT • Have OCTET .The most STABLE group; exist ALONE in nature • Exception to the OCTET is He (only has 2 valence e-) • https://www.youtube.com/watch?v=1PSzSTilu_s • ASAP SCIENCE
Practice • Name the two properties of nonmetals that make them unsuitable for use in electrical wiring. • Use the list of elements belwo to answer the questions
Aim 3 • To identify the trends within the shape of the periodic table.
electronegativity • This is the desire for an atom to GAIN ELECTRONS to gain a full octet • Going ACROSS A PERIOD, electronegativity INCREASES
NONMETALSGAIN e- to achieve full OCTET!HIGH electronegativity values (seen on table S) Bullies • METALS LOSE e- to achieve full OCTET!LOW electronegativity values (seen on table S) Losers
Values range from 0.0 to 4.0 on Table S in your CRT • Most electronegative element: with a value of • Least electronegative elements: with values of
reactivity • ABILITY or TENDENCY of an element to go through a CHEMICAL change (or REACT with another element) • METALS: (recall: the most reactive metal is FRANCIUM) • Going across a period, reactivity DECREASES • Reason: Metals get more stable, moving further from Francium • Going down a group, reactivity INCREASES • Reason: • Radius is bigger, easier to remove an electron. Moving closer to Francium
NONMETALS: (recall: the most reactive nonmetal is FLUORINE) • Going across a period, reactivity INCREASES (until you get to the NOBLE GASES → unreactive) • Reason: Moving closer to Fluorine, closer to the full octet • Going down a group, reactivity DECREASES • Reason: • Moving farther from Fluorine, increased shielding.