Neutralization Reactions in Acids and Bases
90 likes | 105 Vues
Learn about the chemical reaction between acids and bases, resulting in salts and water. Explore titration as an analytical method for determining solution concentrations and equivalence points.

Neutralization Reactions in Acids and Bases
E N D
Presentation Transcript
III. Titration Ch. 19 - Acids & Bases C. Johannesson
A. Neutralization • Chemical reaction between an acid and a base. • Products are a salt (ionic compound) and water. C. Johannesson
A. Neutralization ACID + BASE SALT + WATER HCl + NaOH NaCl + H2O strong strong neutral HC2H3O2 + NaOH NaC2H3O2 + H2O weak strong basic • Salts can be neutral, acidic, or basic. • Neutralization does not mean pH = 7. C. Johannesson
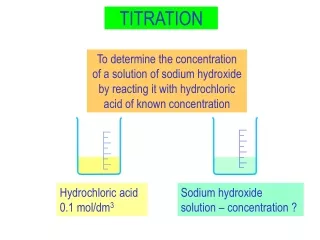
standard solution unknown solution B. Titration • Titration • Analytical method in which a standard solution is used to determine the concentration of an unknown solution. C. Johannesson
B. Titration • Equivalence point (endpoint) • Point at which equal amounts of H3O+ and OH- have been added. • Determined by… • indicator color change • dramatic change in pH • NOTE: end point and eq. pt are not the same! • End point depends on the indicator used C. Johannesson
B. Titration moles H3O+ = moles OH- MVn = MVn M: Molarity V: volume n: # of H+ ions in the acid or OH- ions in the base C. Johannesson
B. Titration C. Johannesson
B. Titration C. Johannesson
B. Titration • 42.5 mL of 1.3M KOH are required to neutralize 50.0 mL of H2SO4. Find the molarity of H2SO4. H3O+ M = ? V = 50.0 mL n = 2 OH- M = 1.3M V = 42.5 mL n = 1 MAVAnA = MBVBnB M(50.0mL)(2) =(1.3M)(42.5mL)(1) M = 0.55M H2SO4 C. Johannesson