The d -Block Elements
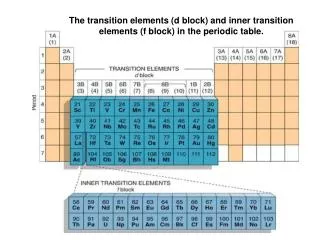
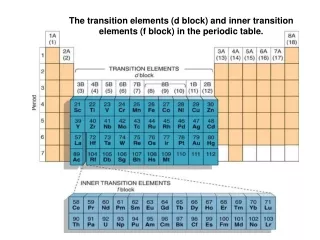
The d -Block Elements. Introduction. d -block elements locate between the s -block and p -block known as transition elements occur in the fourth and subsequent periods of the Periodic Table. period 4. period 5. period 6. period 7. d-block elements. 3d 0. 3d 10.

The d -Block Elements
E N D
Presentation Transcript
Introduction • d-block elements • locate betweenthe s-block andp-block • known as transition elements • occur in thefourth and subsequent periods of the Periodic Table
period 4 period 5 period 6 period 7 d-block elements
3d0 3d10 Introduction Transition elements are elements that contain an incomplete d sub-shell (i.e. d1 to d9) in at least one of the oxidation states of their compounds.
Introduction Sc and Zn are not transition elements because They form compounds with only one oxidation state in which the d sub-shell are NOT imcomplete. Sc Sc3+ 3d0 Zn Zn2+ 3d10
Introduction Cu+ 3d10 not transitional Cu Cu2+ 3d9 transitional
The first transition series the first horizontal row of the d-block elements
Characteristics of transition elements (d-block metals vs s-block metals) • Physical properties vary slightly with atomic number across the series (cf. s-block and p-block elements) • Higher m.p./b.p./density/hardness than s-block elements of the same periods. • Variable oxidation states (cf. fixed oxidation states of s-block metals)
Characteristics of transition elements 4. Formation of coloured compounds/ions (cf. colourless ions of s-block elements) 5. Formation of complexes 6. Catalytic properties
Electronic Configurations The building up of electronic configurations of elements follow: Aufbau principle Pauli exclusion principle Hund’s rule
Electronic Configurations • 3d and 4s sub-shells are very close to each other in energy. • Relative energy of electrons in sub-shells depends on the effective nuclear charge they experience. • Electrons enter 4s sub-shell first • Electrons leave 4s sub-shell first
Cu Cu2+ Relative energy levels of orbitals in atom and in ion
Electronic Configurations • Valence electrons in the inner 3d orbitals • Examples: • The electronic configuration of scandium: 1s22s22p63s23p63d14s2 • The electronic configuration of zinc: 1s22s22p63s23p63d104s2
Electronic configurations of the first series of the d-block elements
A half-filled or fully-filled d sub-shell • has extra stability
d-Block Elements as Metals • d-Block elements are typical metals Physical properties of d-Block elements : good conductors of heat and electricity hard and strong malleableand ductile
d-Block Elements as Metals • Physical properties of d-Block elements: lustrous high melting points and boiling points • Exceptions : Mercury • lowmelting point • liquid at room temperature and pressure
d-Block Elements as Metals • d-block elements • extremely useful as constructionmaterials • strongandunreactive
cheap d-Block Elements as Metals • Iron used for construction and making machinery nowadays abundant easy to extract
d-Block Elements as Metals • Iron corrodes easily often combined with other elements to form steel harder and more resistant to corrosion
d-Block Elements as Metals • Titanium Corrosion resistant, light, strong and withstand large temperature changes used to make aircraft and space shuttles expensive
d-Block Elements as Metals • The similar atomic radii of the transition metals facilitate the formation of substitutional alloys • the atoms of one element toreplacethose of another element • modify their solid structures and physical properties
d-Block Elements as Metals • Chromium • confersinertness to stainless steel • Manganese • confershardness&wearing resistanceto its alloys e.g. duralumin : alloy of Al with Mn/Mg/Cu
Atomic Radii and Ionic Radii • Two features can be observed: • 1. The d-block elements have smalleratomic radii than the s-block elements 2. The atomic radii of the d-block elements do not show much variation across the series
Atomic Radii and Ionic Radii Variation in atomic radius of the first 36 elements
(i) (ii) (i) > (ii) (ii) > (i) On moving across the Period, (i) Nuclear charge (ii) Shielding effect (repulsion between e-)
Atomic Radii and Ionic Radii • At the beginning of the series • atomic number • effective nuclear charge • the electron clouds are pulledcloser to the nucleus • atomic size
In the middle of the series more electrons enter the inner 3d sub-shell The inner 3d electrons shield the outer 4s electrons effectively the effective nuclear charge experienced by 4s electrons increasesvery slowly only a slow decrease in atomic radius in this region
Atomic Radii and Ionic Radii • At the end of the series • the screening and repulsive effects of the electrons in the 3d sub- shell become even stronger • Atomic size
Comparison of Some Physical and Chemical Properties between the d-Block and s-Block Elements • Many of the differences in physical and chemical properties between the d-block and s-block elements • explained in terms of their differences in electronic configurations and atomic radii
1. Density Densities (in g cm–3) of the s-block elements and the first series of the d-block elements at 20C
1. Density • d-block >s-block • the atoms of the d-block elements 1. are generally smaller in size • 2. aremore closely packed • (fcc/hcp vs bcc in group 1) • 3. have higher relative atomic masses
1. Density • The densities • generally increase across the first series of the d-block elements • 1. general decrease in atomic radius across the series • 2. general increase in atomic mass across the series
2. Ionization Enthalpy K Ca (sharp ) ; Ca Sc (slight )
2. Ionization Enthalpy Sc Cu (slight ) ; Cu Zn (sharp )
2. Ionization Enthalpy • The first ionization enthalpies of thed-block elements • greater than those of the s-block elements in the same period of the Periodic Table • 1. The atoms of the d-block elements are smaller in size • 2. greater effective nuclear charges
Sharp across periods 1, 2 and 3 Slight across the transition series
2. Ionization Enthalpy • Going across the first transition series • the nuclear charge of the elements increases • additional electrons are added to the ‘inner’ 3d sub-shell
2. Ionization Enthalpy • The screening effect of the additional3d electrons is significant • The effective nuclear charge experienced by the 4s electrons increases very slightly across the series • For 2nd, 3rd, 4th… ionization enthalpies, • slight and gradual across the series are observed.
3d5 Fe3+ 3d5 Mn2+ 3d5 Cr+ Electron has to be removed from completely filled 3p subshell 3d10 d10/s2
2. Ionization Enthalpy • The first few successive ionization enthalpies for the d-block elements • do not show dramatic changes • 4s and 3d energy levels are close to each other
1541 1668 1910 1907 1246 1538 1495 1455 1084 419 3. Melting Points and Hardness d-block >> s-block 1. both 4s and 3d e- are involved in the formation of metal bonds 2. d-block atoms are smaller
1541 1668 1910 1907 1246 1538 1495 1455 1084 419 3. Melting Points and Hardness K has an exceptionally small m.p. because it has an more open b.c.c. structure.
Sc Ti V Cr Mn Fe Co Ni Cu Zn 1541 1668 1910 1907 1246 1538 1495 1455 1084 419 Unpaired electrons are relatively more involved in the sea of electrons
Sc Ti V Cr Mn Fe Co Ni Cu Zn 1541 1668 1910 1907 1246 1538 1495 1455 1084 419 3d 4s Sc Ti V • m.p. from Sc to V due to the of unpaired d-electrons (from d1 to d3)
Sc Ti V Cr Mn Fe Co Ni Cu Zn 1541 1668 1910 1907 1246 1538 1495 1455 1084 419 3d 4s Fe Co Ni 2. m.p. from Fe to Zn due to the of unpaired d-electrons (from 4 to 0)
Sc Ti V Cr Mn Fe Co Ni Cu Zn 1541 1668 1910 1907 1246 1538 1495 1455 1084 419 3. Cr has the highest no. of unpaired electrons but its m.p. is lower than V. 3d 4s Cr It is because the electrons in the half-filled d-subshell are relatively less involved in the sea of electrons.
Sc Ti V Cr Mn Fe Co Ni Cu Zn 1541 1668 1910 1907 1246 1538 1495 1455 1084 419 4. Mn has an exceptionally low m.p. because it has the very open cubic structure. Why is Hg a liquid at room conditions ? All 5d and 6s electrons are paired up and the size of the atoms is much larger than that of Zn.