Strategies for Electrophilic Aromatic Substitution in Substituted Aromatics
This document explores the nuances of Electrophilic Aromatic Substitution (EAS) reactions, focusing on regioselectivity influenced by steric and electronic effects. It discusses the synthesis of compounds like m-Bromoacetophenone and m-Nitroacetophenone, emphasizing the importance of the order of substituent introduction to achieve desired orientation. Key factors include the activity of substituents and the challenges posed by deactivated aromatics in Friedel-Crafts reactions. The synthesis pathways illustrate how strategic planning can lead to successful product formation in aromatic chemistry.


Strategies for Electrophilic Aromatic Substitution in Substituted Aromatics
E N D
Presentation Transcript
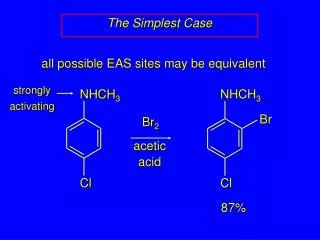
NHCH3 NHCH3 Br Cl Cl The Simplest Case all possible EAS sites may be equivalent strongly activating Br2 aceticacid 87%
CH3 HNO3 H2SO4 C(CH3)3 When activating effects are similar... CH3 substitution occurs ortho to the smaller group NO2 C(CH3)3 88%
CH3 CH3 HNO3 H2SO4 CH3 CH3 NO2 Steric effects control regioselectivity whenelectronic effects are similar position between two substituents is lastposition to be substituted 98%
Br O CCH3 Synthesis of m-Bromoacetophenone If bromine is introduced first, p-bromoacetophenone is major product. para meta
Br O CCH3 O O CH3COCCH3 O CCH3 Synthesis of m-Bromoacetophenone Br2 AlCl3 AlCl3
Factors to Consider order of introduction of substituents to ensure correct orientation Friedel-Crafts reactions (alkylation, acylation) cannot be carried out on strongly deactivated aromatics
NO2 O CCH3 Synthesis of m-Nitroacetophenone If NO2 is introduced first, the next step (Friedel-Crafts acylation) fails.
O CCH3 O O CH3COCCH3 O CCH3 Synthesis of m-Nitroacetophenone O2N HNO3 H2SO4 AlCl3
Factors to Consider order of introduction of substituents to ensure correct orientation Friedel-Craftsreactions (alkylation, acylation) cannot be carried out on strongly deactivated aromatics sometimes electrophilic aromatic substitution must be combined with a functional group transformation
CO2H CH3 CH3 NO2 Synthesis of p-Nitrobenzoic Acid from Toluene Which first? (oxidation of methyl group or nitration of ring)
CO2H CH3 CH3 NO2 Synthesis of p-Nitrobenzoic Acid from Toluene nitration givesm-nitrobenzoicacid oxidation givesp-nitrobenzoicacid
CO2H CH3 CH3 NO2 NO2 Synthesis of p-Nitrobenzoic Acid from Toluene HNO3 Na2Cr2O7, H2O H2SO4, heat H2SO4