OXIDATION PROCESSES IN DRINKING WATER TREATMENT (EXAMPLES)
430 likes | 930 Vues
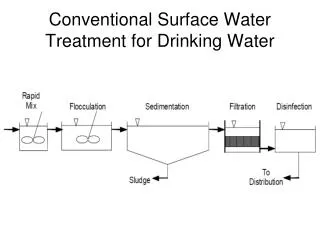
OXIDATION PROCESSES IN DRINKING WATER TREATMENT (EXAMPLES). What processes can we use?. 1. oxidation-reduction. 2. pH and buffering capacity adjustment (pH optimum of processes). 3. chemical precipitation (dissolved particulate ). 4. adsorption (bind to surface).

OXIDATION PROCESSES IN DRINKING WATER TREATMENT (EXAMPLES)
E N D
Presentation Transcript
What processes can we use? 1. oxidation-reduction 2. pH and buffering capacity adjustment (pH optimum of processes) 3. chemical precipitation (dissolved particulate) 4. adsorption (bind to surface) 5. phase separation (removal of different phases: solid/liquid and gas/liquid phase separation) 6. other (e.g. reverse osmosis)
What processes can we use? 1. oxidation-reduction 3. chemical precipitation (dissolved particulate) 4. adsorption (bind to surface) 5. phase separation (removal of different phases: solid/liquid and gas/liquid phase separation) 6. other (e.g. reverse osmosis) 2. pH and buffering capacity adjustment (pH optimum of processes)
Oxidation and reduction The processes are parallel with each other, they take place at the same time The oxidation agent will be reduced (it gets electron), while the reduction agent will be oxidized (it loses electron) In drinking water treatment chemicals are oxidized in order to make them non-soluble, in order to make them less toxic or in order to kill bacteria (disinfection) Oxidizing agents are: oxygen, ozone, chlorine, potassium permanganate, chlorine, chlorine-dioxide, chloramines…
Oxidation is also used to oxidize some compounds to make them non-soluble (oxidation of iron, oxidation of manganese) To convert the soluble compounds into non-soluble compounds: Fe(II) Fe(III) Mn(II) Mn(IV)
Oxidation by air/oxygen Reaction of oxygen with dissolved iron: Fe2+ + 2H2O Fe(OH)2 + 2H+ Fe(II) Soluble form 4Fe(OH)2 + 2 H2O + O2 4Fe(OH)3 Fe(II)Fe(III) Non-soluble form
Oxidation by air/oxygen Reaction of oxygen with dissolved manganese: Mn2+ + 2H2O Mn(OH)2 + 2H+ Mn(II) Soluble form 2Mn(OH)2 + O2 2MnO(OH)2 Non-soluble formMn(II) Mn(IV) MnO(OH)2 MnO2 + H2O
Oxidation by air/oxygen Oxidation of hydrogen-sulfide: 2H2S + O2 2S + 2H2O H2S: rotten-egg odour
Oxidation by ozone Oxidation of: iron Fe(II) Fe(III) manganese Mn(II) Mn(IV) arsenic As(III) As(V) organic compounds compouds causing colour compounds causing taste certain micropollutants (cyanide pollution in 2000) microorganisms
3 O2 + energy 2 O3 Generation: O3 air oxygen from the preparation of feed-gas is needed before ozonation
water is a common medium for microorganisms the spread of infectious diseases (pollution from waste water) The most important aspect of water treatment is the removal of pathogenic microorganisms! This process is called is disinfection and it is usually an oxidation process (expect the UV radiation) Another purpose of oxidation... the disinfection
Disinfectants: ChlorineCl2 ChloraminesNH2-Cl Chlorine dioxideClO2 OzoneO3 SilverAg UV radiation
from the 1910s: the application of chlorine (cheap, efficient) Aim of disinfection 1970s: harmful by-products of chlorine were discovered Disinfection to decrease the number of pathogenic microorganisms in water: bacteria, viruses, protozoa evaluation of other disinfectants
Oxidation by chlorine hypochlorous acid Cl2 + H2O HOCl + H+ + Cl- hypochlorite ion HOCl H+ + OCl- Cl2 Chlorine it reacts with the water: HOCl is more efficient than OCl- the best pH values: between 2 and 6, but this is too low (corrosive water)!! pH is between 7 and 7.5 !!
Oxidation by chlorine Reaction of chlorine with water Cl2 + H2O HOCl + H+ + Cl- Reaction of calcium-hypochlorite with water Ca(OCl)2 + 2H2O 2HOCl + Ca(OH)2 Reaction of sodium-hypochlorite with water NaOCl + H2O HOCl + NaOH formation of hypochlorous-acid (HOCl) !! Addition of Cl2, Ca(OCl)2, NaOCl
Oxidation by chlorine NH3 + HOCl NH2Cl + H2O monochloramine NH2Cl + HOCl NHCl2 + H2O dichloramine NHCl2 + HOCl NHCl3 + H2O trichloramine Breakpoint chlorination Chlorine reacts with ammonia to form chloramines:
Oxidation by chlorine Disadvantage of chlorination Formation of trihalo-methane compounds and halogenated organic compounds Examples: chloroform chlorinated phenol
Oxidation by chlorine Summary of advantages & disadvantages of chlorination Advantages strong oxidant easy to prepare (from NaCl) cheap safe sufficient residual can be maintained Disadvantages reaction with ammonium formation of harmful by-products
Oxidation by chlorine-dioxide ClO2- formation of chlorithe ClO3- formation of chlorathe (harmful) Chlorine-dioxide ClO2 Advantages no reaction with ammonium no THM formation Disadvantages By-products: formation of aromatic chlorinated compounds it has to be produced on-site because of the risk of explosure
Decomposition of ozone – ion-free water T = 20 ˚C pH = 6-7 concentration of ozone (mg/l) time (min) Decomposition of ozone – tap water T = 10 ˚C pH = 8,6 concentration of ozone (mg/l) time (min)
Oxidation by ozonation Secondary disinfectant O3 o o o o o o o o o Disadvantages Advantages It decompoeses easily; There is no residual disinfectant Efficient disinfectant THMs are not formed By-product formation; easily biodegradable organic compounds
Oxidation by ozonation O3 o o o o o o o o o Secondary disinfectant BAC Disadvantages Advantages It decompoeses easily; There is no residual disinfectant Efficient disinfectant THMs are not formed By-product formation; easily biodegradable organic compounds bromate formation Expensive
after treated by ozone after treated by chlorine Regrowth of microorganisms after disinfection 5 4 3 2 1 0 logCFU/mL 0 1 2 3 4 5 6 7 8 9 10 11 time (day) (Miettinen et al.)
APPLICATION OF UV LIGHT FOR DISINFECTION (THIS IS NOT AN OXIDATION PROCESS)
Disinfection by UV radiation A C T G G T C A A C A C G T T G T G A C A C G T UV light – It inactivates the microorganisms by physical way UV 253,7 nm DNA Formation of double bond inhibits replication
Disinfection by UV radiation UV light scatter Particle shading UV light scatter Particle shading UV lamp UV lamp Complete penetration Complete penetration Incomplete penetration Incomplete penetration Region of limited cellular damage Region of limited cellular damage
Disinfection by UV radiation UV light – advantages and disadvantages Advantages It inactivates the microorganisms by physical way Harmful by-products are not formed Short contact time Disadvantages There is no residual disinfectant The impact of water quality on the efficiency of disinfection Buildup of Ca, Mg, Fe scales on the sleeve Biofilm formation on the sleeve Absorption of UV in water; particle interactions
Treated by ozone Without any disinfection g/m2 inner pipe surface Treated by UV radiation Treated by chlorine Biofilm formation (Lund et al.)
Summary, conclusions The importance of the evaluation of water quality parameters The efficiency of disinfection The evaluation of other oxidation processes Formation of by-products Problems with the water quality after disinfection