Comprehensive Guide to Naming Ionic and Covalent Compounds
This guide provides detailed rules for naming ionic and covalent compounds. Ionic compounds consist of a metal and a non-metal, with the metal named first and the non-metal receiving an -ide suffix. Transition metals require charges noted in Roman numerals. For covalent compounds with two non-metals, prefixes indicate the number of atoms involved. Additionally, special rules for naming acids and polyatomic ions are included, distinguishing between binary acids and those containing oxygen. Learn the structure and naming conventions to master chemical nomenclature.

Comprehensive Guide to Naming Ionic and Covalent Compounds
E N D
Presentation Transcript
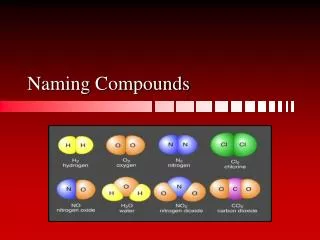
Naming Compounds Ionic Compounds
Always have a METAL and NON-METAL • Metal always named first • Write element name • Non-metal follows after • Add suffix -ide Simple Ionic Compounds
NaCl • Sodium Chloride • MgO • Magnesium Oxide • CaBr2 • Calcium Bromide Simple Ionic Examples
Must include charge of transition metal • Transition metals located in middle d-block • Charge written in () as Roman numerals • Only 2 exceptions • Zinc (2+) Zn • Silver (1+) Ag • Find charge by using known charge of non-metal • I, II, III, IV, V, VI, VII, VIII Transition Metal Ionic
CrO • O has a 2- charge • Cr must have a 2+ charge • Chromium (II) Oxide • AuN • N has a 3- charge • Au must have 3+ charge • Gold (III) Nitride • Cr2O3 • Oxygen has a 2- charge (6- overall) • 2 Cr ions have a 6+ ion Cr has 3+ charge • Chromium (III) Oxide Transition Metal Examples
Calcium chloride • CaCl2 • Barium Sulfide • BaS • Copper (I) Oxide • Cu2O • Iron (II) Nitride • Fe3N2 Naming to Formula
Naming Compounds Covalent Compounds
Covalent compounds involve 2 non-metals • For first non-metal, add prefix to indicate number in compound EXCEPT when there is only 1 • Mono, di, tri, tetra, penta, hexa • For second non-metal, add prefix AND –ide suffix • Mono, di, tri, tetra, penta, hexa Covalent Compounds
N2O5 • DinitrogenPentoxide • CO2 • Carbon Dioxide • CO • Carbon Monoxide Covalent Examples
Carbon Tetrachloride • CCl4 • Sulfur Hexaflouride • SF6 • Video Naming to Formula
Naming Compounds Acids and Polyatomic Ions
Polyatomic Ions can take the place of an anion or cation • Only cation that we use is NH4+, Ammonium Naming Polyatomic Ions
All binary acids (H and 1 other el.) • Include prefix HYDRO- • Followed by anion • Suffix –IC added • Add “acid” to the very end • HCl • Hydrochloric acid Naming Acids
All acids with Oxygen • Identify which polyatomic ion it is • Use polyatomic name • Suffix –ate –ic • Suffix –ite –ous • Add “acid” to very end • HNO3 (nitrate ion) • Nitricacid • HNO2 (nitrite ion) • Nitrousacid Naming Acids