Dental Stem Cells
Dental Stem Cells. Reviewed by Terrell F. Pannkuk, D.D.S. M.Sc.D. Mesenchymal Stem Cells Derived from Dental Tissues vs. Those from Other Sources: Their Biology and Role in Regenerative Medicine. This presentation is an outline derived from the following article:.


Dental Stem Cells
E N D
Presentation Transcript
Dental Stem Cells Reviewed by Terrell F. Pannkuk, D.D.S. M.Sc.D
Mesenchymal Stem Cells Derived from Dental Tissues vs. Those from Other Sources: Their Biology and Role in Regenerative Medicine This presentation is an outline derived from the following article: G. T. –J. Huang, S. Gronthos, and S. Shi, J Dent Res 88 (9): 792-806, 2009
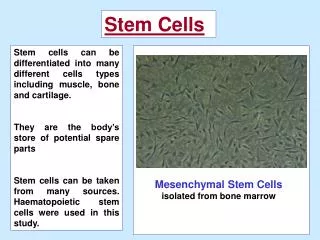
Stem Cells Derived from Dental Tissues • Mesenchymal Stem Cells (MSC’s) • Sources: • Bone Marrow (Friedenstein et al, 1976; Caplan, 1991; Prockop, 1997; Pittenger et al, 1999; Gronthos et al, 2003) • Adipose Tissue/Umbilical Cord (Mareschi et al, 2001; Zuk et al, 2001) • Lineages: • Osteogenic • Chondrogenic • Adipogenic
Other Lineages Possibly Derived from Bone Marrow Mesenchymal Stem Cells Myogenic Neurogenic Tenogenic
Dental Tissue MSC’s • Human Pulp Tissue (DPSC’s, post-natal dental pulp stem cells) • Gronthos et al, 2000 • Exfoliated Deciduous Teeth (SHED) • Miura et al, 2003 • Periodontal Ligament (PDLSC) • Seo et al, 2004 • Apical Papilla (SCAP) • Sonoyama et al, 2006, 2008 • Dental Follicle Precursors (DFPC) • Morsczeck et al, 2005
Dental Stem Cell Lineages • Osteo/Odontogenic • Adipogenic • Neurogenic *Dental Stem cells appear to be more committed to odontogenic paths than BMMSC’s
BMMSC’s • Colony Forming Unit Fibroblasts (CFU-F’s) • Self Renewal (like hematopoietic lines) • 30-50 PD’s (population doublings) • Cell Surface Markers • Heterogeneity supports stromal hierarchy of differentiation • Minor proportion involved with extensive proliferation
Dental MSC’s • Dental tissues are specialized tissues that do not undergo continuous remodeling as shown in bony tissues • Dental mesenchyme is termed ‘ectomesenchyme’ due to its earlier interaction with the neural crest.
Isolation of Dental Pulp Stem Cells • Enzymatically isolated and seeded onto dentin to promote “Odontoblast-like” cells. • Multilineage differentiation of hDPSC subpopulations: • Adipogenic • Neurogenic • Osteogenic • Chondrogenic • Myogenic
Ectopic Formation of Dentin-Pulp-like Complex • Transplanted DPSC’s mixed with hydroxyapatite/tricalcium phosphate (HA/TCP) forms ectopic pulp-dentin like tissue complexes in immunocompromised mice. (Gronthos et al., 2000; Batouli et al., 2003) Odontoblast-like cells express sialophosphoprotein (DSPP), producing dentinal tubules similar to natural dentin
SHED (Exfoliated Deciduous Teeth SC’s) • Fast proliferation • Greater PD (population doubling) • Sphere like cluster formation (cultured neurogenic medium • Also termed “immature stem cells) • Unable to regenerate a complete dentin-pulp complex in vivo • Unlike DPSC’s can differentiate into bone forming cells.
SCAP ( Apical Papilla SC’s) • Odontogenic differentiation • Adipogenic differentiation
DPSC’s vs. SCAP • Apical papilla is a precursor to radicular pulp • Earlier line of stem/progenator cells (SCAP) • SCAP’s superior source of stem cells
PDLSC’s (periodontal ligament sc’s) • Form cementoblasts and osteoblasts • Homeostasis and regeneration of perio tissues • Cementum-PDL structure unique from BMMSC’s and DPSC’s
DFPC’s (Dental Follicle Precursor Cells) • Periodontium, cementum, PDL, alveolar bone precursors • Source: impacted third molars
Dental MSC’s vs. BMMSC’s • Gene Expression: 4000 known human genes • Cooperative regulation of genes for cell signaling, cell communication, or metabolism • BMMSC’s only form bone tissue in mice • DPSC chondrogenic potential is weak • BMMSC’s have stronger adipogenic potential than both DPSC’s and SCAP • Neurogenicity in dental stem cells more potent than BMMSC’s (probably due to neural crest origin)
MSC Niche • Specialized microenvironment needed to maintain stem cells in their multipotent state. (Schofield, 1978) • Considered a fixed compartment: • Regulate proliferation • Control fate of stem cell progeny • Prevent exhaustion and death of stem cells (Scadden, 2006; Jones and Wagers, 2008) • BMMSC niche-perivascular area of bone marrow • DPSC niche-perivascular and perineural sheath areas
MSC Homing • MSC’s in human blood is low under steady state conditions • Ex Vivo expanded MSC’s injected into the blood stream have a limited capacity to home into various tissues and organs. • Injected Ex Vivo-expanded BMMSC’s through intravenous infusion lodge mainly in lungs, smaller amounts in liver, heart, spleen, and damaged areas of the brain. • No evidence that BMMSC’s migrate to orofacial /dental organs
Immunomodulation of MSC’s • Allogenic MSC’s are well tolerated by the recipient hosts (Xenografts do not take). • MSC’s have an immunosuppressive effect • Preliminary study shows interferon may act to differentiate MSC’s into osteoblasts • Inflammatory reactions against scaffold materials and serum components lead to the production of cytokines
Dental MSC-Based Therapy for Regenerative Medicine • SCAP and PDLSC’s for Bio-root Engineering • Single cells from dog tooth buds at the bell stage seeded onto scaffolds and transplanted back into sockets resulted in some dentin structure regeneration with no enamel or root formation (Honda et al., 2006) • Kuo et al., 2007 used pigs, expanded ex vivo expansion of bud cells from bell stage and observed some root structures along with periodontium.
Obstacles to Tooth Regeneration • Abnormal (small) tooth size • Lack of consistent root formation • Incomplete eruption into functional occlusion.
Regeneration of Perio Defects with PDLSC’s • PDGF (platelet derived growth factor) • IGF (insulin derived growth factor) • PRP (platelet rich plasma) • Cell based regenerative therapy: • Ex vivo expanded autologous BMMSC’s facilitated repair of perio defects (Yamada et al., 2006) • PDL regeneration is as important as bone regeneration otherwise ankylosis ensues • rhBMP-2 therapy does not regenerate PDL • PDLSC’s may be an ideal source to regenerate PDL (Liu et al., 2008)
Pulp Tissue Engineering/Regeneration • Early attempts (Myers and Fountain, 1974) allowed a blood clot to form in the canal but only connective tissue formed. • More recently pulp cells grown on polyglycolic acid (PGA) formed pulp-like tissue in vitro and in vivo (Gu et al., 1996; Moony et al., 1996, and Burma et al., 1999) • Since the isolation and characterization of DPSC’s SHED and SCAP, more sophisticated regenerative investigation has occurred (Huang et al., 2006, 2008; Murray et al., 2007; Prescott et al., 2008)
Modern Pulp Regeneration • SHED seeded onto synthetic scaffolds seated into pulp chamber space formed odontoblast-like cells that located against the existing dentin surface. (not orthotopic) (Cordeiro et al., 2008) • Speculation: undifferentiated MSC’s residing in the periapical tissue and BMMSC’s in the alveolar bone of the jaws can be introduced into the root canal space and via blood clots to allow for pulp tissue regeneration and formation of odontoblasts (Myers and Fountain, 1974)
Modern Pulp Regeneration (cont.) • More realistically: the known characteristics of PDLSC’s, DPSC’s, and SCAP suggest that it is unlikely that odontoblasts can be derived from PDL or periapical bone. • When BMMSC’s and DPSC’s are transplanted into the subcutaneous space of immunocompromised mice they form BM-like and Dentin-pulp like complexes respectively (Gronthos et al., 2000) • DPSC’s have shown osteogenic potential but there is no evidence showing BMMSC’s can give rise to functional odontoblasts and dentin.
The Future: • Need to understand mechanisms of self-renewal and regulate stem cell growth to generate sufficient numbers • Need to overcome regulation of differentiation into specific tissue production, specialized extracellular matrices (bone, dentin, cartilage, and tendon). The production of the extracellular matrix and its maturation into specialized tissues involves a sequential activation of cascades of signals. • Need to understand the interactions between stem cells and the immune system. Allogenic dental MSC’s may suppress recipient host short and long term immunorejection. • Controlling and preventing ex vivo expanded MSC’s from transformation . Adipose –derived MSC’s lost genetic stability over time and are prone to tumor formation (Rubio et al., 2005)