Periodic Table
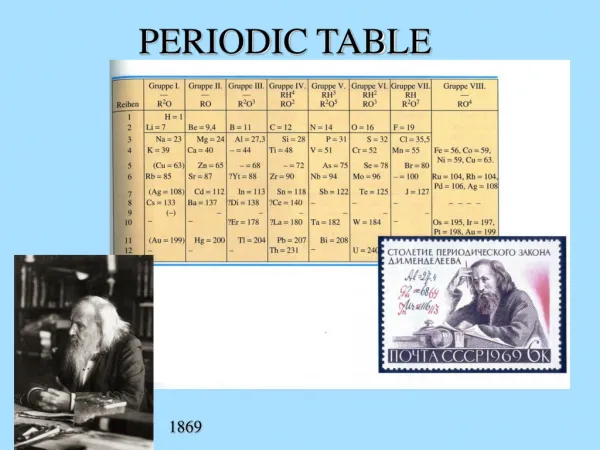
Periodic Table. A Work in Progress. History. 1817 German chemist Johann Dobereiner Triads of similar chemical properties 1864 John Newlands – English Law of Octaves 1869 Dmitri Mendeleev – Russian Cards, left holes where he thought elements should be Dutch Anton van den Broek


Periodic Table
E N D
Presentation Transcript
Periodic Table A Work in Progress
History • 1817 German chemist Johann Dobereiner • Triads of similar chemical properties • 1864 John Newlands – English • Law of Octaves • 1869 Dmitri Mendeleev – Russian • Cards, left holes where he thought elements should be • Dutch Anton van den Broek • arranged according to charge, not mass
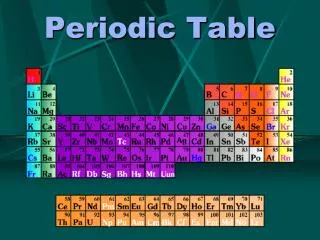
Organization • Periods – ROWS • properties change gradually • Group (family) – COLUMNS • have similar properties
Key to the Periodic Table • Element name • Atomic number (Whole #, element ID) • Symbol • Atomic mass (weighted average of isotopes) • State of matter
Metals • Solids (except mercury) • Luster (shiny) • Conducts heat, electricity • Maleable (easily shaped) • Ductile (can be shaped into wire)
Non-metals • Gases or brittle solids • Poor conductors of electricity • Only 17
Metalloids - Semimetals • Some properties of metals/nonmetals
Groups (Columns) • Representative elements – groups 1-2, 13-18 (some metals, metalloids, non-metals) • Transition elements – groups 3-12 – metals • Inner transition elements – inside bottom 2 rows – lanthanide, actinide series
Group 1 Alkali Metals • Far left column on PT • Silvery solids • Low density • React violently with water, burns skin • Never found in pure form in nature • Forms +1 ions
Group 2 Alkaline Earth Metals • 2nd column from left • Denser, harder, higher melting points • Not as reactive as alkali metals • Burn different colors – fireworks! • Forms +2 ions
Groups 13-16 • BCNO – large family, diverse • Similar bonding characteristics • Group 13 – Boron family • Group 14 – carbon family • Group 15 – Nitrogen group • Group 16 – Oxygen family • Forms -2 ions
Halogens • Group 17 • Forms salts/ -1 ions • Poisonous • Very reactive • Not found in pure form in nature • When bonded to Carbon, destroys ozone layer
Noble Gases • Far right of PT • Gases • Light up when electrical current goes through • Don’t react • Outer energy levels filled • Group 18
Transition Metals • Groups 3-12 • Conduct heat, electricity • Combine with oxygen different ways • Dissolve in acid (except gold) • Higher melting points than other elements
Inner Transition Metals • Lanthanides • Soft, silvery metals • Spark when struck • Actinides • all radioactive • Some synthetic
Energy Levels • 2 • 8 • 8 • 18 • 18 • 32 • 32
Valance Electrons • In outer incomplete energy level • Used to bond with other atoms • Shown in electron dot diagrams