Ionic Equilibria (Acids and Bases)
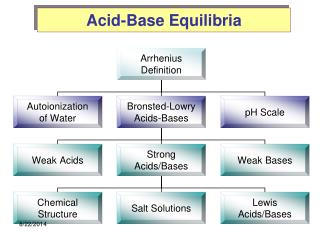
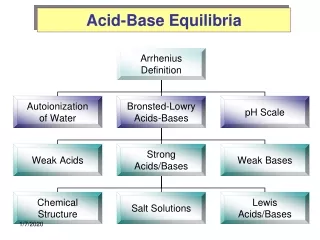
Ionic Equilibria (Acids and Bases). Chapter 18. Phase I. STRONG ELECTROLYTES. Strong Electrolytes. Strong Acids – 7 strong acids Strong Bases – Group 1 and 2 (Ca, Sr, Ba) hydroxides Soluble Salts Some molecular compounds – requires extensive ionization. Calculating Ion Concentration.

Ionic Equilibria (Acids and Bases)
E N D
Presentation Transcript
Ionic Equilibria (Acids and Bases) Chapter 18
Phase I STRONG ELECTROLYTES
Strong Electrolytes • Strong Acids – 7 strong acids • Strong Bases – Group 1 and 2 (Ca, Sr, Ba) hydroxides • Soluble Salts • Some molecular compounds – requires extensive ionization
Calculating Ion Concentration • Calculate the concentration of each ion and the pH in 0.050M HNO3 HNO3(aq) + H2O (l) H30+ (aq) + NO3-(aq) • Assume 100% dissociation (all strong electrolytes) • H30+ (aq) = 0.050M • NO3-(aq) = 0.050M • pH = -log(H30+) = -log(0.050) = 1.30
Calculating Ion Concentration • Calculate the concentraion of ions and the pH of 0.020M Ba(OH)2 solution. Ba(OH)2 Ba2+ + 2OH- • Assume strong electrolyte = 100% dissociation • Ba2+ = 0.020M • OH- = 2(0.020M) = 0.040M • [OH-][H30+] = 1 x 10-14 • [H30+] = 2.5 x 10-13 • pH = -log(2.5 x 10-13) = 12.60
Auto-ionization of Water 2H2O (l) H3O+ (aq) + OH- (aq) Kc = [H3O+][OH-] *temp dependent Kc = Kw = [H3O+][OH-] = 1.0 x 10-14 @ 25oC Table 18-2 (p755) has values at different temperatures
Auto-ionization of Water • Calculate the concentration of H3O+ and OH- in 0.050M HCl HCl + H2O H3O+ + Cl- • H3O+ = 0.050M • [H3O+][OH-] = 1.0 x 10-14 • [0.050M][OH-] = 1.0 x 10-14 • [OH-] = 2.0 x 10-13
Question • Why don’t we add in the concentration of H3O+ from the auto ionization of water? • Compare 10-7 to 0.50…no contest • So much H3O+ that equilibrium is pushed to H2O
pH and pOH • pH and pOH express the acidity and basicity of dilute solutions
Cool relationship between pH and pOH [H3O+][OH-] = 1.0 x 10-14 log[H3O+] + log[OH-] = log(1.0 x 10-14) and -log[H3O+] - log[OH-] = -log(1.0 x 10-14) pH + pOH = 14.00
Example Problem • Calculate [H3O+] , pH, pOH, and [OH-] for 0.020M HNO3. HNO3 + H2O H3O+ + NO3- • [H3O+] = [0.020] • pH = -log(0.020) = 1.70 • pOH = 14 – pH = 14 – 1.70 = 12.30 • [OH-] = 10-pOH = 10-12.30 = 5.0 x 10-13
Phase II WEAK ELECTROLYTES
Ionization of Weak Acids • The activity of water is assumed to be 1 as it is nearly a pure liquid with this weak ionization.
Ionization of Weak Acids • Small ka = weak acid • Large ka = strong acid • Ionization constants are measured experimentally • Freezing point depression • Electrical conduction measurement • pH measurements
Example • In a 0.12M solution of weak monoprotic acid (HA) is 5.0% dissociated, calculate ka. HA H+ + A-
Example • The pH of weak acid H is measured at 2.97 in a 0.10M solution. Calculate the Ka.
Example • Calculate the concentration of all species in a 0.15M acetic acid solution if Ka = 1.8 x 10-5 • When x is <5% of the number being added or subtracted from the initial concentration, it may be NEGLECTED. • % Ionization = [CH3COOH ionized] [CH3COOH initial]
Ionization of Weak Bases Review Appendix G, p. A-15
Example • Calculate the concentration of various species in 0.15M NH3 and % Ionization.
Example • An Ammonia solution has a pH of 11.37. Calculate its molarity.
Polyprotic Acids • Acids with more than one proton • Most are considered weak acids, however the first proton ionizes the strongest • Typically 104 to 106 difference between Ka
Example • H3AsO4, arsenic acid • Ka1 = 2.5 x 10-4 • Ka2 = 5.6 x 10-8 • Ka3 = 3.0 x 10-13 • Calculate the concentration of all species in 0.100M solution of H3AsO4.
Solvolysis • Solvolysis – the reaction of a dissolved substance with a solvent • Hydrolysis – the reaction of a dissolved substance with WATER. • Generally, anions of STRONG acids do not hydrolyze • Generally, anions of WEAK acids do hydrolyze
Solvolysis KCl + H2O NR (no pH change) Neither K nor Cl will Hydrolyze (from strong sources) NaClO + H2O HClO + Na+ + OH- • Anions of weak acids are relatively strong bases. • Kb = [HClO][OH-] [ClO-]
Solvolysis • Works for all conjugate acid/base pairs in water.
Salts Where things get murkey Yay!
Salts • Objective: • What happens to pH when I put a salt of a Strong Acid, Weak Acid, Strong Base, or Weak Base into water?
Cation From: Strong Base (Na+) Na+ + H2O NR No Effect Anion From: Strong Acid (Cl-) Cl- + H2O NR No Effect Strong Acid and Strong Base
Cation From: Strong Base (K+) K+ + H2O NR No Effect Anion From: Weak Acid (ClO-) ClO- + H2O HClO + OH- Increases pH Strong Base and Weak Acid
Cation From: Weak Base (NH4+) NH4+ + H2O H3O+ + NH3 Decreases pH Anion From: Strong Acid (NO3-) NO3- + H2O NR No Effect Weak Base and Strong Acid
Cation From: Weak Base (NH4+) NH4+ + H2O H3O+ + NH3 Decreases pH Anion From: Weak Acid (ClO-) ClO- + H2O HClO + OH- Increases pH Weak Base and Weak Acid
Weak Base and Weak Acid • How do you know if the pH will increase or decrease? • If Kb = Ka, then pH = 0 • If Kb > Ka, then pH = + • If Kb < Ka, then pH = -