General, Organic, and Biochemistry, 7e
440 likes | 635 Vues
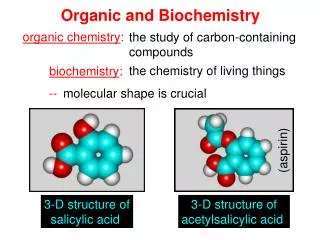
General, Organic, and Biochemistry, 7e. Bettelheim, Brown, and March. Chapter 3. Chemical Bonds. Atoms and Their Ions. In 1916, Gilbert N. Lewis pointed out that the lack of chemical reactivity of the noble gases indicates a high degree of stability of their electron configurations.

General, Organic, and Biochemistry, 7e
E N D
Presentation Transcript
General, Organic, and Biochemistry, 7e Bettelheim, Brown, and March
Chapter 3 Chemical Bonds
Atoms and Their Ions • In 1916, Gilbert N. Lewis pointed out that the lack of chemical reactivity of the noble gases indicates a high degree of stability of their electron configurations
The Octet Rule • Octet rule: the tendency of group 1A-7A elements to react in ways that achieve an outer shell of eight valence electrons • an atom that loses one or more electrons becomes a negatively charged ion called an anion • an atom that gains one or more electrons becomes a positively charged ion called a cation
The Octet Rule • Example: in losing one electron, a sodium atom forms a sodium ion, which has the same electron configuration as neon Na (11 electrons): 1s2 2s2 2p63s1 Na+ (10 electrons): 1s2 2s3 2p6
The Octet Rule • Example: in gaining one electron, a chlorine atom forms a chloride ion, which has the same electron configuration as argon chlorine atom (17 electrons): 1s2 2s2 2p63s23p5 chloride ion (18 electrons): 1s2 2s2 2p63s23p6
The Octet Rule • The octet rule gives us a good way to understand why Group 1A-7A elements form the ions they do; but it is not perfect • ions of period 1 and 2 elements with charges greater than +2 are unstable. For example, boron does not lose its three valence electrons to become B3+, nor does carbon lose its four valence electrons to become C4+ • ions of period 1 and 2 elements with charges greater than -2 are also unstable. For example, carbon does not gain four valence electrons to become C4- • the octet rule does not apply to Group 1B-7B (transition elements), most of which form ions with two or more different positive charges
Naming Cations • Elements of Groups 1A, 2A, and 3A form only one type of cation; the name of the cation is the name of the metal followed by the word “ion”
Naming Cations • For cations derived from transition and inner transition elements, most of which form more than one type of cation, • use either Roman numerals to show charge, or • use the suffix -ous to show the lower positive charge and the suffix -ic to show the higher positive charge
Naming Anions • For monatomic (containing only one atom) anions, add “ide” to the stem part of the name • here are the monatomic anions we deal with most often
Polyatomic Ions • common names, where still widely used, are given in parentheses
Forming Chemical Bonds • According to the Lewis model • an atom may lose or gain enough electrons to acquire a filled valence shell and become an ion. An ionic bond is the result of the force of attraction between a cation and an anion. • an atom may share electrons with one or more other atoms to acquire a filled valence shell. A covalentbond is the result of the force of attraction between two atoms that share one or more pairs of electrons.
Electronegativity • Electronegativity: a measure an atom’s attraction for the electrons it shares in a chemical bond with another atom • on the Pauling scale, fluorine, the most electronegative element is assigned a value of 4.0, and all other elements are assigned values relative to fluorine
Ionic Compounds • According to the Lewis model, an ionic bond is formed by the transfer of one or more valence electrons from an atom of lower electronegativity to an atom of higher electronegativity • the more electronegative atom gains one or more valence electrons and becomes an anion • the less electronegative atom loses electrons and becomes a cation • the compound formed by the combination of an anion and a cation is called an ionic compound
Forming an Ionic Bond • In forming sodium chloride, NaCl • we use a single-headed curved arrow to show this transfer of one electron
Formulas of Ionic Compounds • The total number of positive charges must equal the total number of negative charges • lithium ion and bromide ion form LiBr • barium ion and iodide ion form BaI2 • aluminum ion and sulfide ion form Al2S3 • sodium ion and bicarbonate ion form NaHCO3 • potassium ion and phosphate ion form K3PO4
Naming Ionic Compounds • Binary ionic compounds • the name of metal from which the positive ion is formed followed by the name of the negative ion; subscripts are ignored • AlCl3 is aluminum chloride • LiBr is lithium bromide • Ag2S is silver sulfide • MgO is magnesium oxide • KCl is potassium chloride
Naming Ionic Compounds • Binary ionic compounds of metals that form two different cations • for systematic names, use Roman numerals to show charge on the metal ion; for common names, use the -ous, -ic suffixes • CuO is copper(II) oxide; cupric oxide • Cu2O is copper(I) oxide; cuprous oxide • FeO is iron(II) oxide; ferrous oxide • Fe2O3 is iron(III) oxide; ferric oxide
Naming Ionic Compounds • Ionic compounds that contain polyatomic ions • name the positive ion first followed by the name of the negative ion • NaNO3 is sodium nitrate • CaCO3 is calcium carbonate • NaH2PO4 is sodium dihydrogen phosphate • NH4OH is ammonium hydroxide • FeCO3 is iron(II) carbonate; ferrous carbonate • Fe2(CO3)3 is iron(III) carbonate; ferric carbonate • CuSO4 is copper(II) sulfate; cupric sulfate
Forming a Covalent Bond • A covalent bond is formed by sharing one or more pairs of electrons • the pair of electrons is shared by both atoms and, at the same time, fills the valence shell of each atom • example: in forming H2, each hydrogen contributes one electron to the single bond
Polarity of Covalent Bonds • Although all covalent bonds involve sharing of electron pairs, they differ in the degree of sharing • nonpolar covalent bond: electrons are shared equally • polar covalent bond: electron sharing is not equal • the degree of sharing depends on the relative electronegativities of the atoms in the bond
Polarity of Covalent Bonds • Examples:
Polarity of Covalent Bonds • In a polar covalent bond • the more electronegative atom gains a greater fraction of the shared electrons and acquires a partial negative charge; indicated by d- or the head of a crossed arrow • the less electronegative atom has a lesser fraction of the shared electrons and acquires a partial positive charge; indicated by d+ or the tail of a crossed arrow
Drawing Lewis Structures 1. Determine the number of valence electrons in the molecule 2. Decide on the arrangement of atoms in the molecule 3. Connect the atoms by single bonds 4. Show bonding electrons as a single line; show nonbonding electrons as a pair of Lewis dots 5. In a single bond, atoms share one pair of electrons; in a double bond, they share two pairs, and in a triple bond they share three pairs.
Lewis Structures • Examples: (the number of valence electrons is given in parentheses after the molecular formula
Lewis Structures • Examples • draw a Lewis structure for hydrogen peroxide, H2O2 • draw a Lewis structure for methanol, CH3OH • draw a Lewis structure for acetic acid, CH3COOH
Exceptions to the Octet Rule • Atoms of period 2 elements use 2s and 2p orbitals for bonding • these four orbitals can contain a maximum of 8 electrons; hence the octet rule • Atoms of period 3 elements have one 3s orbital, three 3p orbitals, and five 3d orbitals • these nine orbitals can accommodate more than eight electrons; period 3 atoms can have more than eight electrons in their valence shells
Exceptions to the Octet Rule • Phosphorus • Sulfur
Molecular Compounds • Molecular compound: a compound in which all bonds are covalent • Naming binary molecular compounds • the less electronegative element is named first (it is generally written first in the formula) • prefixes “di-”, tri-”, etc. are used to show the number of atoms of each element; the prefix “mono-” is omitted when it refers to the first atom, and is rarely used with the second atom. Exception: carbon monoxide • NO is nitrogen oxide (nitric oxide) • SF2 is sulfur difluoride • N2O is dinitrogen oxide (laughing gas)
VSEPR Model • Valence-Shell Electron-Pair (VSEPR) Model • valence electrons of an atom may be involved in forming single, double, or triple bonds, or they may be unshared • each combination creates a negatively charged region of electron density around the nucleus • because like charges repel each other, the various regions of electron density around an atom spread so that each is as far away from the others as possible
VSEPR Model • Predict the shape of methane, CH4 • the Lewis structure shows carbon surrounded by four regions of electron density • according to the VSEPR model, the four regions radiate from carbon at angles of 109.5°, and the shape of the molecule is tetrahedral • the measured H-C-H bond angles are 109.5°
VSEPR Model • Predict the shape of ammonia, NH3 • the Lewis structure shows nitrogen surrounded by four regions of electron density; three regions contain single pairs of electrons, and the fourth contains an unshared pair of electrons • according to the VSEPR model, the four regions radiate from nitrogen at angles of 109.5°, and the shape of the molecule is pyramidal • the measured H-N-H bond angles are 107.3° • the unshared pair is not shown on this model
VSEPR Model • Predict the shape of water, H2O • the Lewis structure shows oxygen surrounded by four regions of electron density; two regions contain single pairs of electrons, and the third and fourth contain unshared pairs of electrons • according to the VSEPR model, the four regions radiate from oxygen at angles of 109.5°, and the shape of the molecule is bent • the measured H-O-H bond angle is 104.5° • the unshared pairs are not shown on the model
VSEPR Model • Predict the shape of formaldehyde, CH2O • the Lewis structure shows carbon surrounded by three regions of electron density; two regions contain single pairs of electrons and the third contains two pairs of electrons forming the double bond to oxygen • according to the VSEPR model, the three regions radiate from carbon at angles of 120°, and the shape of the molecule is planar (trigonal planar) • the measured H-C-H bond angle is 116.5°
VSEPR Model • Predict the shape of ethylene, C2H4 • the Lewis structure shows carbon surrounded by three regions of electron density; two regions contain single pairs of electrons and the third contains two pairs of electrons forming the double bond to the other carbon • according to the VSEPR model, the three regions radiate from carbon at angles of 120°, and the shape of the molecule is planar (trigonal planar) • the measured H-C-H bond angle is 117.2°
VSEPR Model • Predict the shape of acetylene, C2H2 • the Lewis structure shows carbon surrounded by two regions of electron density; one region contains a single pair of electron, and the second contains three pairs of electrons forming the triple bond to carbon • according to the VSEPR model, the two regions radiate from carbon at an angle of 180°, and the shape of the molecule is linear • the measured H-C-C bond angle is 180°
Polarity of Molecules • A molecule will be polar if • it has polar bonds, and • its centers of partial positive and partial negative charges lie at different places within the molecule • Carbon dioxide, CO2, has two polar bonds but, because of its geometry, is a nonpolar molecule
Polarity of Molecules • Water, H2O, has two polar bonds and, because of its geometry, is a polar molecule
Polarity of Molecules • Ammonia, NH3, has three polar bonds and, because of its geometry, is a polar molecule
Polarity of Molecules • Both dichloromethane, CH2Cl2, and formaldehyde, CH2O, have polar bonds and are polar molecules
Chemical Bonds End Chapter 3