E5 GL evolves from a Conjecture (or a grand excuse) to a Solid Ground
30 likes | 144 Vues
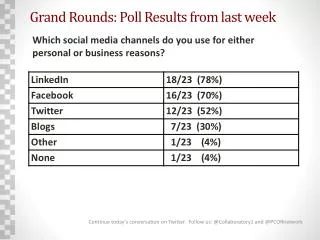
The E5 guideline has transitioned from a conjecture to a solid foundation for drug approval, thanks to early decision-makers who embraced risk based on expertise. Though initially unproven, the E5 guideline evolves with experience and real-world data, as demonstrated by the approval rates of new chemical entities (NCEs) in Japan from 1999 to 2001. This evolution emphasizes the importance of public health as a central concern for regulators, reflecting the balance between efficiency in drug development and societal needs.

E5 GL evolves from a Conjecture (or a grand excuse) to a Solid Ground
E N D
Presentation Transcript
E5 GL evolves from a Conjecture (or a grand excuse) to a Solid Ground • E5 guideline is something like a conjecture that has not been proved yet. • I am sincerely grateful to those who made the first “let’s sink or swim” decision, based on their expertise. • Must be carefully applied. • It does evolve, however, with experiences, as mentioned in E5 GL itself. • Making it to a solid ground is our real goal. Kitasato-Harvard 2002 S ONO
Drugs approved in Japan based on the “E5 conjecture” • 1999: 2 / 39 NCEs approved • 2000: 3 / 39 NCEs approved • 2001: 5 / 23 NCEs approved • It is these drugs that will establish the scientific authenticity of E5 guideline. • Real-world constatation has just started. Kitasato-Harvard 2002 S ONO
Public Health is THE concern for the regulators. • Public health is not A concern; it is THE concern for regulatory people fueled by taxpayers’ money. • If their conservativeness ‘truly’ came from such concern, who can blame them? (Unrealistic assumption?) • Efficiency on producer’s (ie, investigators, industry) side does not necessarily guarantee efficiency in society; consumers (ie, regulators, practitioners, patients) have their say. (as long as economists at Harvard School of Public Health taught me right....) Kitasato-Harvard 2002 S ONO