Advantages of Bivalirudin Monotherapy Over Heparin/Enoxaparin with GP IIb/IIIa Inhibitors
170 likes | 287 Vues
Bivalirudin monotherapy demonstrates significant advantages over traditional anticoagulants like heparin and enoxaparin, particularly in reducing major bleeding incidents while offering comparable ischemic protection. This treatment enhances net clinical outcomes, showcasing its effectiveness in acute coronary syndrome management. The synthesis of direct thrombin inhibition with lower bleeding risks and simpler administration offers a favorable alternative for patient care, especially in the context of contemporary interventional cardiology practices.

Advantages of Bivalirudin Monotherapy Over Heparin/Enoxaparin with GP IIb/IIIa Inhibitors
E N D
Presentation Transcript
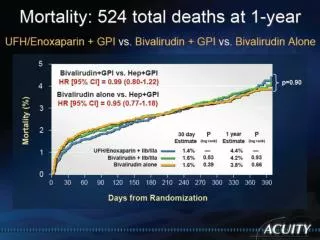
Bivalirudin Advantage Compared to Heparin/Enoxaparin with GP IIb/IIainhibitors,Bivalirudinmonotherapy significantly reduces major bleeding while providing similar ischemic protection, and improves net clinical outcome.
IIa C AT IIa S Hep UFH Direct antithrombin AT Xa AT Xa Pentasaccharide LMWH
Fondaparinux: ASynthetic Factor Xa Inhibitor Intrinsic pathway Extrinsic pathway Antithrombin Xa Xa AT AT AT Fondaparinux THROMBIN II IIa Fibrinogen Fibrin clot Adapted with permission from Turpie AGG et al. NEngl J Med. 2001;344:619.
IXa VIIIa Ca2+ PL Xa Va Ca2+ PL Key Steps in Coagulation Pathway Intrinsic pathway Intrinsic pathway Extrinsic pathway 1 Inhibition of one molecule of factor Xa can inhibit the generation of 50 molecules of thrombin2 X Xa 50 IIa II Fibrin Fibrinogen 1. Rosenberg RD, Aird WC. N Engl J Med 1999;340(20):1555–64. 2. Wessler S, Yin ET. Thrombo Diath Haemorrh 1974;32(1):71–8. Clot
Fondaparinux:A Synthetic Inhibitor of Factor Xa • Once daily administration • Rapid onset (Cmax/2=25 min) • Half life: 15-18 h. • Effects reversible with administration of activated Factor VII (Novoseven®) • No liver metabolism • Renal clearance • No protein binding (other than AT) • No reported cases of HIT • No dose adjustment necessary in elderly Herbert JM et al. Cardiovasc Drug Rev. 1997;15:1. van Boeckel CAA et al. Angew Chem, Int EdEngl. 1993;32:1671.
OASIS-6: Randomized, Double Blind 12,000 Patients with STEMI < 12 h of symptom onset Inclusion: ST 2 mm prec leads or 1 mm limb leads Exclusion: Contra-ind. for anticoagulant, INR>1.8, pregnancy, ICH<12 mo. Lytics (SK, TPA, TNK, RPA), Primary PCI or no reperfusion (eg. late) Stratification UFH not indicated UFH indicated Randomization Randomization Fondaparinux 2.5 mg Placebo Fondaparinux 2.5 mg UFH JAMA 2006;295:1519-30
Primary Efficacy OutcomeDeath/MI at 30 Days 0.12 UFH/Placebo 0.10 Fondaparinux 0.08 0.06 Cumulative Hazard HR 0.86 95% CI 0.77-0.96 P=0.008 0.04 0.02 0.0 0 3 6 9 12 15 18 21 24 27 30 Days The OASIS-6 Trial Group. JAMA 2006;295:1519-30
Death or MI 3 or 6 months 0.12 UFH/Placebo 0.10 Fondaparinux 0.08 0.06 Cumulative Hazard HR 0.88 95% CI 0.79-0.99 P=0.029 0.04 0.02 0.0 0 18 36 54 72 90 108 126 144 162 180 Days The OASIS-6 Trial Group. JAMA 2006;295:1519-30
Primary: Efficacy: Death, MI, refractory ischemia 9 day • Safety: Major bleeds • Risk benefit: Death, MI, refractory ischemia, major bleeds • Secondary: Above & each component (especially deaths) at 30 & 180 d • Hypothesis: First test non-inferiority, then test superiority
Death at 6 Months Enoxaparin 0.06 Fondaparinux 0.04 Cumulative Hazard HR 0.89 95% CI 0.79-0.99 p=0.037 0.02 0.0 0 20 40 60 80 100 120 140 160 180 Days
Death or MI: 6 Months Enoxaparin 0.12 0.10 Fondaparinux 0.08 Cumulative Hazard 0.06 HR 0.91 95% CI 0.84-0.99 p=0.036 0.04 0.02 0.0 0 20 40 60 80 100 120 140 160 180 Days
Major Bleeding: 6 Months 0.06 Enoxaparin 0.05 0.04 Fondaparinux 0.03 Cumulative Hazard HR 0.72 95% CI 0.63-0.82 p<<0.00001 0.02 0.01 0.0 0 20 40 60 80 100 120 140 160 180 Days
Death, MI, RI or MajorBleedingat 6 Months Enoxaparin 0.15 Fondaparinux 0.10 Cumulative Hazard HR 0.87 95% CI 0.81-0.93 p<<0.00001 0.05 0.0 0 20 40 60 80 100 120 140 160 180 Days
Fondaparinux Advantages Disadvantages • Difficult to monitor (no aPTT or ACT) • Long half-life • Catheter thrombosis during PCI • SC administration • Potential exists for outpatient management • Once-daily administration • Predictable anticoagulant response • Fixed dose • No antigenicity • Potentially no need for serologic parameters • Does not cross the placenta • HIT antibodies do not cross-react • Decreased bleeding complications vs UFH or LMWH Simoons ML, et al. J Am Coll Cardiol. 2004;43:2183-2190.Yusuf S, et al. N Engl J Med. 2066;354:1464-1476.