Entropy (S)
Entropy (S). A measure of the amount of disorder of a system Nature tends to move towards more disorder (higher entropy) Which has the greater entropy in each pair? NH 3 (g) or NH 3 (l) NH 3 (g) or NH 3 ( aq ) CO 2 (s) or CO 2 (g) KBr (s) or KBr (l).

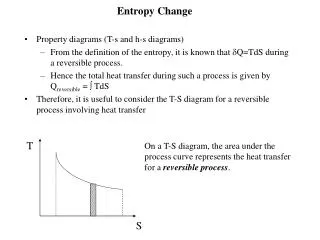
Entropy (S)
E N D
Presentation Transcript
Entropy (S) • A measure of the amount of disorder of a system • Nature tends to move towards more disorder (higher entropy) Which has the greater entropy in each pair? • NH3(g) or NH3(l) • NH3(g) or NH3(aq) • CO2(s) or CO2(g) • KBr(s) or KBr(l)
Does entropy increase or decrease for each of the following? • C2H5OH(s) C2H5OH(l) • H2O(g) H2O(l) • CH3OH(l) CH3OH(g) • NH3(l) NH3(s)
Calculate S for reaction • Calculate S just like you calculate H = sum of all products – sum of all reactants using S values • S – (+) = entropy increases (more disorder) (-) = entropy decreases (more order/less disorder) Determine S for the reaction 2NiS(s) + 3 O2(g) 2SO2(g) + 2NiO(s) using the standard entropy values.
Gibb’s Free Energy - G • Determines the spontaneity of a chemical reaction • Calculate two ways: 1. sum of all products – sum of all reactants 2. G = H - TS • G - (-) = spontaneous reaction (0) = equilibrium (+) = not spontaneous
Using the information you have available, calculate H, S, and G for the reaction 2SO2(g) + O2(g) 2SO3(g) at 250C and 1 atm. • Calculate G for 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(g) using the free energies of formation values.
G = -RTlnK • Consider the following reaction at 25.00C: 2NO2(g) N2O4(g). The values of H and S are -58.03 kJ/mol and -176.6 J/K*mol, respectively. Calculate the value of K. • Consider the auto ionization of water. H2O(l) H+(aq) + OH-(aq). At 400C, the Kw = 2.92x10-14. Calculate G.