Acid-Base Titration
Acid-Base Titration. Acid-Base Titration. Acid-base titration is a laboratory procedure used to determine – among other things – the unknown concentration of an acid or a base. Acid-Base Titration.

Acid-Base Titration
E N D
Presentation Transcript
Acid-Base Titration • Acid-base titration is a laboratory procedure used to determine – among other things – the unknown concentration of an acid or a base.
Acid-Base Titration • During an acid-base titration, an acidic solution of known concentration is added drop by drop to a base solution of unknown concentration (or vise versa) until all the hydrogen ions (H1+) have exactly neutralized all the hydroxide ions (OH1-). An appropriate acid-base indicator will signal... ...when moles H1+ equals moles OH1-. HOH OH1- H1+ HOH HOH H1+ OH1- H1+ HOH HOH HOH OH1- H1+ H1+ HOH OH1- HOH OH1- HOH
Acid-Base Titration • During an acid-base titration, an acidic solution of known concentration is added drop by drop to a base solution of unknown concentration (or vise versa) until all the hydrogen ions (H1+) have exactly neutralized all the hydroxide ions (OH1-). Count the number of H1+ ions added. How many OH1- ions were present in the original solution? HOH H1+ HOH HOH H1+ H1+ HOH HOH HOH HOH H1+ H1+ H1+ six HOH HOH HOH
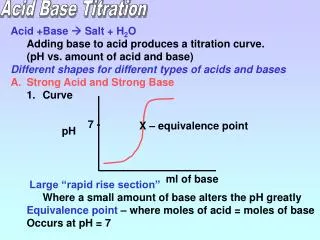
Acid-Base Titration • Remember, neutralization occurs when... the moles of H1+ from the acid = the moles of OH1- from the base • The point of exact neutralization is called the equivalence point and is signaled by a color change (end point) in an acid-base indicator.
Acid-Base Titration OH 1) How many moles of NaOH does it take to neutralize 1.5 mol HCl? ________ 2) How many moles of LiOH does it take to neutralize 1.5 mol H2SO4? ________ 3) How many moles of Ba(OH)2 does it take to neutralize 1.5 moles of HNO3? ________ 4) How many moles of Ca(OH)2 does it take to neutralize 1.5 moles of H2CO3? ________ H 1.5 mol OH H2 3.0 mol (OH)2 H 0.75 mol (OH)2 H2 1.5 mol
Acid-Base Titration • Once the titration is complete, the concentration of the unknown solution is determined by employing the titration formula: MAVA = MBVB Beware: • MA = molarity of H1+ but VA = volume of the acid! • MB = molarity of OH1- but VB = volume of the base! this equation is tricky! MA H1+ VA acid MB OH1- VB base
Acid-Base Titration • The molarity of H1+ is equal to the acid’s molarity times the number of H1+’s on the acid. MA = #H1+ x Macid • The molarity of OH1- is equal to the base’s molarity times the number of OH1-’s on the base. MB = #OH1- x Mbase
Acid-Base Titration M of H1+ 5) If the MA of an H2SO4 solution is 2.0 M, then what is molarity (M) of the H2SO4 solution? MA = #H1+ x Macid 1 H2SO4 can produce 2 H1+! 2.0 M = (2) (x) 1.0 M H2SO4 x =
Acid-Base Titration 6) If the molarity (M) of an HNO3 solution is 1.0 M, then what is the MA of the solution? MA = #H1+ x Macid M of H1+ 1 HNO3 can produce 1 H1+! x = (1) (1.0 M) x = 1.0 M H1+
Acid-Base Titration M of H1+ 7) If the MA of an H3PO4 solution is 1.2 M, then what is molarity (M) of the H3PO4 solution? MA = #H1+ x Macid 1 H3PO4 can produce 3 H1+! 1.2 M = (x) (3) x = 0.40 M H3PO4
Acid-Base Titration 8) If the molarity (M) of an Ba(OH)2 solution is 0.25 M, then what is the MB of the solution? MB = #OH1- x Mbase M of OH1- 1 Ba(OH)2 can produce 2 OH1-! (2) (0.25 M) x = 0.50 M OH1- x =
Acid-Base Titration M of OH1- 9) If the MB of an Ca(OH)2 solution is 1.0 M, then what is molarity (M) of the Ca(OH)2 solution? MB = #OH1- x Mbase 1 Ca(OH)2 can produce 2 OH1-! 1.0 M = (x) (2) x = 0.50 M Ca(OH)2
Acid-Base Titration 10) If the molarity (M) of an NaOH solution is 0.12 M, then what is the MB of the solution? MB = #OH1- x Mbase M of OH1- 1 NaOH can produce 1 OH1-! x = (1) (0.12 M) x = 0.12 M OH1-
Acid-Base Titration 11) How many milliliters of a 1.50 M solution of H2SO4 would exactly neutralize 7.50 mL of a 0.250 M solution of Ba(OH)2? MAVA = MBVB (7.50 mL) (0.500 M) (x) = (3.00 M) x = 1.25 mL Choose an appropriate acid-base indicator. __________________ methyl orange or bromcresol green
Acid-Base Titration 12) If 20.0 mL of an unknown solution of HCl is titrated with a 2.0 M solution of NaOH such that it required 37.0 mL of the NaOH to reach the end point, what is the molarity of the HCl? MAVA = MBVB (2.0 M) (20.0 mL) = (37.0 mL) (x) x = 3.7 M of H1+ Choose an appropriate acid-base indicator. __________________ The molarity of the HCl = 3.7 M litmus or bromthymol blue
Acid-Base Titration 13) If 150.0 mL of 0.050 M H2CrO4 solution is titrated against a 0.30 M NaOH solution, then how many milliliters of the base would be needed to reach the end point? MAVA = MBVB (0.30M) (150.0 mL) = (0.100 M) (x) x = 50. mL Choose an appropriate acid-base indicator. __________________ thymol blue or phenolphthalein
Acid-Base Titration 14) What is the molar concentration of a 25.0 mL sample of NH3 (NH4OH) if it takes 8.0 mL of a 0.15 M solution of HBr to neutralize the NH3 (NH4OH)? MAVA = MBVB (x) (25.0 mL) (8.0 mL) = (0.15 M) x = of OH1- 0.048 M Choose an appropriate acid-base indicator. __________________ The molarity of the NH3 = 0.048 M methyl orange or bromcresol green
Acid-Base Titration 15) How many milliliters of 0.10 M nitric acid (HNO3) would be needed to neutralize 16.0 mL of a 0.15 M solution of Ba(OH)2? MAVA = MBVB (0.30 M) (x) = (0.10 M) (16.0 mL) x = 48 mL Choose an appropriate acid-base indicator. __________________ methyl orange or bromcresol green
Acid-Base Titration 16) A student neutralized 16.4 milliliters of HCl by adding 12.7 milliliters of 0.620 M KOH. What was the molarity of the HCl acid? MAVA = MBVB (0.620 M) (16.4 mL) = (x) (12.7 mL) x = 0.480 M of H1+ Choose an appropriate acid-base indicator. __________________ The molarity of the HCl = 0.480 M litmus or bromthymol blue
Acid-Base Titration 17) What is the molarity of a solution containing 20.0 grams of NaOH in 500. milliliters of solution? M = mol L # mol = given mass GFM x = __________ x = __________ 0.500 mol 20.0 g 40.0 g/mol 0.500 L x = 0.500 mol x = 1.00 M