Acid Base Titration
Acid Base Titration. Titration. the process of determining the molarity of an acid or base by using an acid-base reaction where one reactant is of known molarity . Get your lab book and copy the lab. Title : Acid/Base Titration

Acid Base Titration
E N D
Presentation Transcript
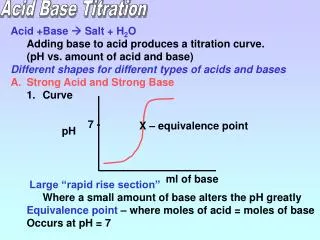
Titration • the process of determining the molarity of an acid or base by using an acid-base reaction where one reactant is of known molarity.
Get your lab book and copy the lab. Title: Acid/Base Titration Purpose: The purpose of this lab is to practice safe lab technique, to expand our understanding of the acid/base chemical reaction, and to gain experience in the use of indicators. Materials: 250 mL flask 50 mL beaker for 1.0 M HCl (hydrochloric acid) 50 mL beaker to measure water 2 eye droppers water supply phenolphthalein indicator unknown concentration NaOH (sodium hydroxide) solution
Procedure: 1. Place 20 mL of water in a clean 250 mL flask. 2. Place 1 mL of 1.0 M HCl in the flask. 3. Place 5 drops of phenolphthalein indicator in the flask and swirl. 4. Begin adding drops, one at a time of Unknown concentration NaOH to the flask. Be sure and COUNT the number of drops that you add. You will be looking for a COLOR CHANGE. SWIRL THE FLASK EACH TIME YOU ADD A DROP. 5. When the color change is stable, stop adding drops. 6. Perform steps #1 - #5 two more times. 7. Average your results. 8. Calculate the molarity of the unknown. 9. Calculate the percentage error of your results.
Data: Volume of 1.0 M HCl = 1 mL Number of drops of NaOH needed to reach the endpoint =______ Volume of 1.0 M HCl = 1 mL Number of drops of NaOH needed to reach the endpoint =______ Volume of 1.0 M HCl = 1 mL Number of drops of NaOH needed to reach the endpoint =______ Average number of drops = _____________
Calculations: Write the balanced equation for the reaction and determine the mole ratio. NaOH + HCl _______ + _______ Calculate the average number of mL of NaOH needed to reach the endpoint based on 20 drops = 1.0 mL. (circle your answer) Calculate the molarity of the unknown NaOH solution using Ma*Va = Mb*Vb. (circle your answer) Calculate the percentage error of your results using the provided expression. (circle your answer). Tell what you learned during the lab and provide any explanations of what errors may have occurred.
Calculations: Calculate the percentage error of your results using the provided expression. (circle your answer). Tell what you learned during the lab and provide any explanations of what errors may have occurred.