Periodic Table
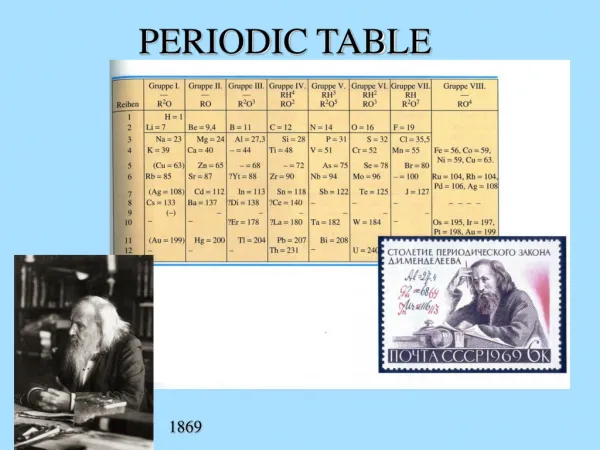
The Periodic Table, formulated by Russian chemist Dmitri Mendeleev in 1869, organizes elements based on their physical and chemical properties. This system categorizes elements by periodic properties that repeat every seven elements in horizontal rows (periods) and vertical columns (groups). Key elements include hydrogen, carbon, and iron, each critical to various chemical reactions and essential for life. From highly reactive gases like oxygen to metals used in construction, understanding the periodic table is fundamental to chemistry and science.

Periodic Table
E N D
Presentation Transcript
Periodic Table • Russian chemist Dmitri Mendeleev in 1869 and b • The Periodic Table of Elements is used to organize elements based on • physical properties such as • hardness (brittle or malleable) • conductivity (thermal and electrical) • chemical properties such as • reactivity • flammability
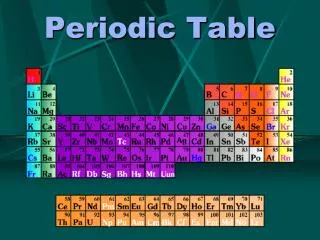
Periodic Properties of the Elements Periodic describes something that occurs in regular intervals. The elements properties repeat every seven elements
The rows are read left to right • The horizontal rows are called periods • Row # = # of energy levels in electron cloud • Lower rows = larger atoms (bigger diameter) • Atomic mass increases from left to right across a period.
Vertical columns are called groups or families • Column # (group #) = # of valence electrons • Elements in same group have similar chemical and physical properties
M E T a L L O I D S Non Me t a l S Metals, Non-Metals and Metalloids ME T A L S
Five Elements to Know: H C O Al Fe
Oxygen • Gas at room temp. • Highly reactiveelement! • Combines easily with other elements to form compounds called “oxides”: iron oxide (rust), copper oxide, carbon dioxide. • At high enough temp, many substances react violently with oxygen - combustion (burning) • Used by most organisms for cellular respiration. • Makes up 21 % of earths atmosphere.
Is a gas Simplest and most abundant element in universe Scientists estimate ¾ of universe is hydrogen! Stars made of mostly hydrogenUsed as rocket fuelHIGHLY FLAMMABLE and REACTIVE
The Hindenberg Disaster • May 6, 1937 in Lakehurst NJ. • German airship bursts into flames trying to land. • It was filled with hydrogen gas. • Completely incinerated in just over 30 seconds. • 62 out of 97 people survived.
Is a non metal • Mostly obtained from coaldeposits. • Black soot from burning wood is mostly carbon. • Graphite - form of carbon used in pencils. • Diamond– • Carbon +pressure+ heat + time = Diamond
Combines with other elements to make organic compounds like proteins, fats, and carbohydrates. • Most important element for living things!
One of most abundant of all metals. • Used to make steel (iron + carbon). • Helps to oxygenate the blood and makes it red. • Iron is a magnetic metal. • Good conductor. “Iron Age” approx. 1200 B.C. - 300 B.C.
Very conductive metal. • Lightweight (low density) but strong. • Used in cans, foil and kitchen utensils, parts of airplanes and cars.