Molarity Calculations
Molarity Calculations. Calculations – Part 1. Molarity of solution from the mass of solute and volume of solution. How to make a specific volume of a given concentration (M). Concentration of ions in a solution of strong electrolytes Moles of solute present in a given volume of solution

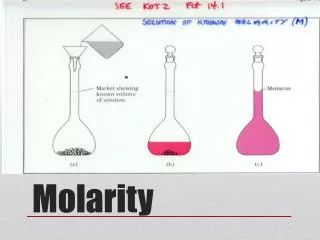
Molarity Calculations
E N D
Presentation Transcript
Calculations – Part 1 • Molarity of solution from the mass of solute and volume of solution. • How to make a specific volume of a given concentration (M). • Concentration of ions in a solution of strong electrolytes • Moles of solute present in a given volume of solution • How to dilute solutions to a specific concentration.
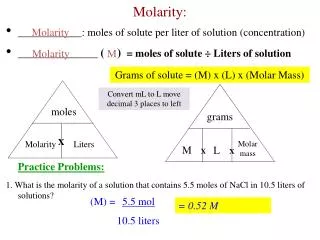
What is the molarity of a solution made by dissolving 15.0 g of NaOH in enough water to make 350.0 mL of solution?
What are the concentrations of ions present in a 0.25 M AlCl3 solution?
DilutionC1V1 = C2V2 • What is the concentration of a solution made by diluting 25.0 mL of 6.0 M HCl to a total volume of 500.0 mL?
Dilution • Describe how to make 250.0 mL of 0.250M NaOH from a 6.00 M stock solution.
Solution Stoichiometry Mg + 2 HCl MgCl2 + H2 • What volume of 1.50 M HCl is needed to fully react 0.40 grams of Mg?
Acid Base Titrations 2 NaOH + H2SO4 Na2SO4 + 2 H2O • The complete reaction of 7.00 mL of H2SO4 requires 23.50 mL of 0.250 M NaOH. • How many moles of NaOH were needed to react the H2SO4? • How many moles of H2SO4 were neutralized? • What is the concentration of the H2SO4? (the real question)