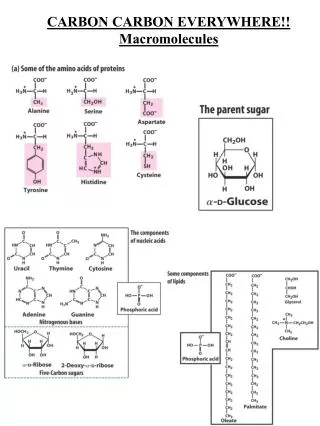
Essential Facts About Carbon: Nature, Uses, and Properties
Carbon, symbol C, is a non-metal and the sixth element on the periodic table, known since prehistoric times. It has a melting point of 3652°C and a boiling point of 4827°C. Carbon appears in various forms, including amorphous, diamond, and graphite, playing a vital role in life processes. It forms from natural substances and is crucial in both biological and industrial applications. However, certain carbon compounds can be harmful to living cells. For detailed insights, visit various chemistry resources.

Essential Facts About Carbon: Nature, Uses, and Properties
E N D
Presentation Transcript
Carbon By: Matthew – 7.4
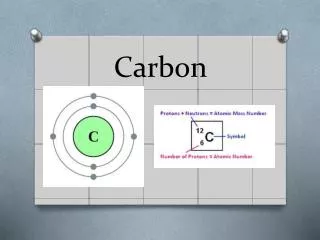
Some Helpful Information • Name: Carbon • Symbol : C • Number of Atomic: 6 • Melting point: 3652 °C - 3820 (K) • Boiling point: 4827 °C - 5100 (K) • Is it a metal?: No, it is not a metal.
Discovery of Carbon Date of Discovery: Carbon is known from a Latin word “Carbo” which was discovered during the prehistoric time. Discoverer: There are no discoverer because Carbon are made from natural things.
Descriptions of the Element • Carbon is made and found free in the nature • Carbon are forming with limited applications used • Thousands of Carbon are used in life processses • Carbon has three forms: - amorphous (lampblack), diamond and graphite -It produces a lot of things such as a diamond that can be sold with a high amount of money
Pictures http://chemistry.about.com/od/elementfacts/a/carbon.htm http://chemistry.about.com/od/elementfacts/a/carbon.htm
The Bad/Dangers Things • Many of the compounds kill the living cells • Plants and Animals are combine with compounds such as the Carbon, hydrogen, oxygen, etc • The remains of the animals and plants are forming deposits • Some animals and plants are killed
Uses of Carbon • Carbon can form to with limit applications • Thousands of Carbon can be used in the life processes • The Three forms can really helped people and they are worth of money such as the diamond
Interesting Facts • The origin name are ‘Carbon' comes from the Latin word carbo, for charcoal. • Carbon is made from free of nature and has been known since prehistoric time. • Carbon is made in the interiors of stars.
Helpful Information • Crystal Structure: Hexagonal • Density: 2.25 (graphite) • Lattice Structure: Diagonal • Specific Heat (@20°C J/g mol):0.711 • Magnetic Ordering: diamagnetic
Bibliography Helmenstine, Anne Marie. “Carbon Chemical & Physical properties.” Carbon Facts. 25 April 2010 <http://chemistry.about.com/od/element facts/a/carbon.htm> Helmenstine, Anne Marie. “Carbon – The Chemical Basis for Life.” 10 Carbon Facts. 25 April 2010 <http://chemistry.about.com/od/element facts/a/carbon.htm> BV, Lenntech. “Carbon – C.” Carbon. 25 April 2010 <http://www.lenntech.com/periodic/element/c.htm>