Electrolytes
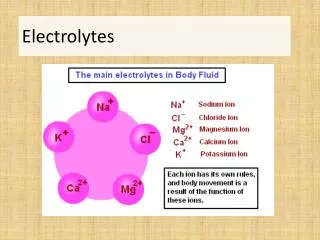
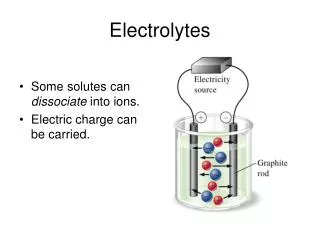
Electrolytes. Electrolytes. Are substances that form positive(+) and negative (-) ions in water Conduct an electric current. Electrolytes in the Body. Carry messages send to and from the brain as electrical signals Maintain cellular function with the correct concentrations electrolytes.

Electrolytes
E N D
Presentation Transcript
Electrolytes • Are substances that form positive(+) and negative (-) ions in water • Conduct an electric current
Electrolytes in the Body • Carry messages send to and from the brain as electrical signals • Maintain cellular function with the correct concentrations electrolytes
Strong Electrolytes are 100% ionized saltsH2O 100%ions NaCl(s) Na+(aq) + Cl-(aq) H2O CaBr2(s) Ca2+(aq) + 2Br- (aq) acidsH2O HCl(g) H3O+(aq) + Cl-(aq)
Weak electrolytes • Most of the solute exists as molecules in solution • Only a few solute particles produce ions HF + H2O H3O+(aq) + F- (aq) acid NH3 + H2O NH4+(aq) + OH- (aq) base
Classification of electrolytes • Acids Binary acids • Ternary acids (oxyacids) • Thioacids • Monoprotic acids • Polyprotic acids • Strong acids • Weak acids
Bases Monovalent • Polyvalent • Strong • Weak • Basic oxides – base anhydrides • Oxides of metals • Salts Strong electrolytes!!!
Important Facts • HCl, HNO3 – strong acids • 100% dissociation (no equilibrium, no K) • HCl H+ + Cl– • 0%HCl, 100% H+, 100% Cl– • H+ is a proton, a subatomic particle and does not exist alone in solution • Really H+ + H2O H3O+ (hydronium ion) HNO3 + H2O NO3– + H3O+
conc. of ionic form a = total concentration a ≤1 Strong electrolytes weak electrolytes Degree of dissociation: a Strong electrolyte: a1 Weak electrolyte: a < 0.5
_ + H H H H+ Cl- H _ O O + H O O H H H Conductivity of electrolytes
H O Na+OH- H O H H H O O H H H 1 Conductivity= + 1 cm 1 cm ( = resistance) V = 1 cm3
H2SO4 NH4Cl NaCl cc. =specific conductivity
v a = inf Equivalent conductivity: Conductivity of equivalent amount of electrolyte = V V = equivalent dilution in cm3 ainf = 1 v = a
400 300 200 100 cc. ( ) 1 10-4 10-5 10-1 10-2 10-3 10 Strong acid Strong base Weak acid Weak base Salt