FGFR
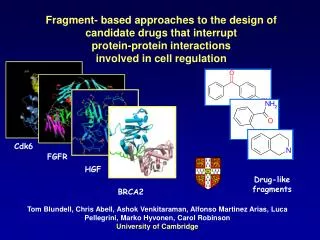
S161. H114. E115. K91. Q173. R93. D171. Fragment- based approaches to the design of candidate drugs that interrupt protein-protein interactions involved in cell regulation. Cdk6. FGFR. HGF. Drug-like fragments. BRCA2.

FGFR
E N D
Presentation Transcript
S161 H114 E115 K91 Q173 R93 D171 Fragment- based approaches to the design of candidate drugs that interrupt protein-protein interactions involved in cell regulation Cdk6 FGFR HGF Drug-like fragments BRCA2 Tom Blundell, Chris Abell, Ashok Venkitaraman, Alfonso Martinez Arias, Luca Pellegrini, Marko Hyvonen, Carol Robinson University of Cambridge
Many difficult targets are found amongst multiprotein complexes of cell signalling and regulation Further complication: often weak binary complexes lead to stable multiprotein complexes.
Traditional use of X-ray and modelled 3D structures Structural biology and Drug Discovery Target Selection Lead Discovery Lead Development Target Identification & Validation Screening Hits-to-leads Lead optimisation Sequence-structure homology recognition Structural genomics Structure-based Screening
Complex of cyclin D-dependent kinase Cdk6 bound to the cell cycle inhibitor p19INK4d. Large, flat and uninteresting: difficult to bind small molecules? Brotherton, Dhanaraj, Blundell, Laue, Nature 395, 244-250, 1998
STRATEGIES FORLEAD DISCOVERY WITH TARGETS THAT ARE MULTIPROTEIN COMPLEXES • Exploitation of hot spots in protein-protein interaction surfaces. • Formation of large aggregates of hydrophobic or amphipathic molecules – a specific mechanism of non-specific inhibition! • Exploitation of allosteric effects. • Fragment –based appoaches
Multiprotein Complexes Preformed globular structures Discontinuous epitopes + Preformed but adaptive globular structures + One or more partners become structured on assembly Continuous epitopes +
RAD51 is an essential enzyme in DNA repair. • The breast cancer susceptibility protein BRCA2 controls RAD51 activity via a direct protein-protein interaction. • Disruption of the BRCA2-RAD51 binding interface blocks RAD51 activity.
Expression and purification of RAD51 for lead identification. Screening: • NMR (in vitro) • X-ray (in crystal)
b a d c e f Figure 10 a, 986677. b, 1014147. c, 302916. d, 300842. e, 984356. f, 977759. The compound 977759 would probably be more co-planar than is predicted in the docking solution, due to bond conjugation.
Linking or coupling Alternative Fragment strategies Growing
Novel hitsS1 Neutral Fragment Thrombin fragment identification Novel binding sitesS2-S4 Fragment • S2/S4 fragment potency 10mM • Linked S1 and S2/S4 fragment: new hybrid compound • potency 200nM
Thrombin inhibition S4-pocket S2-pocket Ximelagatran Exanta (AZ) Melagatran S1-pocket • To identify novel, non-peptidic inhibitors which lack strongly basic functionality
Fragment linking: thrombin inhibition S4-pocket S2-pocket S1-pocket
S1 Fragment hits S4-pocket S2-pocket IC50 = 330 mM S1-pocket
S2-S4 Ligands IC50 = 300 mM S4-pocket S2-pocket S1-pocket
Fragment Linking IC50 = 300 mM S4-pocket S2-pocket IC50 = 3.4 nM IC50 = 330 mM S1-pocket theoretical values Kd (additive) 100 nM Kd(superadditive) 0.1 nM
1 1 cocktail of three fragments 0.5 mM + enzyme 20 mM fragment 1 only + enzyme 2 3 1 Biophysical chemist Screen fragments against protein targets using some of the following techniques: (i) X-ray crystallography (ii) NMR spectroscopy (Water LOGSY) (iii) Isothermal titration calorimetry (iv) Surface plasmon resonance (v) Non-covalent mass spectrometry Have recently opened a new facility for Biological Chemistry which includes new ITC and SPR equipment.
Dynamic combinatorial chemistry (DCC) Fragment combination bound to protein target Reactive fragments Fragment combinations Detect product either by Stopping equilibration and identifying major species Direct observation by x-ray crystallography M. S. Congreve et al., Angew. Chemie Int. Ed., 2003, 42, 4479-82
Essential Phe and Ala side-chains PheHisThrAlaSerGly F-H-T-A-S-G
Lead identification • Strategy: Find fragments that bind the Phe or Ala pockets; link them Screen fragment library by mass spec, SPR, etc • Factors: • Leads may have a high MW and/or a low affinity • Kd of the BRC:RAD51 interaction ~1µM in preliminary SPR studies 3. Initial aim: Leads with Kd ~1-10nM & most ‘drug-like’ characteristics
Proof-of-concept & optimization studies for potential leads Inhibition of the BRC peptide-RAD51 interaction in ELISA or FP assays Inhibition of RAD51 focus formation after DNA damage In vitro sensitization of cell lines to DNA damaging agents Increasing potency
Potential clinical profiles • Genetic & cell biological studies demonstrate that disruption of the BRCA2-RAD51 interaction - • Suppresses DNA double-strand break repair by homologous recombination during G2 • Sensitizes cells to DNA cross-linking agents, PARP inhibitors, and replication blockers • So, potential clinical profiles could include - • Combination with radiation or radiomimetics to overcome G2 resistance • Combination with DNA cross-linking agents, PARP poisons or topoisomerase inhibitors
Conventional screens Synthetic libraries [MRCT] Natural product libraries [EU] Lead optimization Pre-clin development Parallel approaches to lead identification Fragment-based screens Lead ID, proof-of-concept studies
Research Scientist Appointments • Medicinal Chemist. • Biophysical Chemist • Molecular biologist/biochemist • X-ray crystallography • Cell biologist for assays
1 1 cocktail of three fragments 0.5 mM + enzyme 20 mM fragment 1 only + enzyme 2 3 1 Biophysical chemist Screen fragments against protein targets using some of the following techniques: (i) X-ray crystallography (ii) NMR spectroscopy (Water LOGSY) (iii) Isothermal titration calorimetry (iv) Surface plasmon resonance (v) Non-covalent mass spectrometry Have recently opened a new facility for Biological Chemistry which includes new ITC and SPR equipment.
Synthetic organic chemist Preparation of fragment library (analysis of chemical structures, knowledge of reactivity, solubility etc) Strategy to build up from initial fragment hits to lead compounds (going from mM to nM) Preparation of successive generations of compounds as go from hits to leads. Compound synthesis, purification and characterisation (specific skil set, cannot be done by e.g. biochemist or molecular biologist) Development of fragment combining approaches (including dynamic combinatorial chemistry and dynamic combinatorial crystallography) We have a recent success at carrying out fragment based drug discovery in the University resulting in nM inhibitor (thrombin project)
Thrombin inhibition S4-pocket S2-pocket Ximelagatran Exanta (AZ) Melagatran S1-pocket • To identify novel, non-peptidic inhibitors which lack strongly basic functionality
Fragment linking: thrombin inhibition S4-pocket S2-pocket S1-pocket
S1 Fragment hits S4-pocket S2-pocket IC50 = 330 mM S1-pocket
S2-S4 Ligands IC50 = 300 mM S4-pocket S2-pocket S1-pocket
Fragment Linking IC50 = 300 mM S4-pocket S2-pocket IC50 = 3.4 nM IC50 = 330 mM S1-pocket theoretical values Kd (additive) 100 nM Kd(superadditive) 0.1 nM
1 1 cocktail of three fragments 0.5 mM + enzyme 20 mM fragment 1 only + enzyme 2 3 1 Biophysical chemist Screen fragments against protein targets using some of the following techniques: (i) X-ray crystallography (ii) NMR spectroscopy (Water LOGSY) (iii) Isothermal titration calorimetry (iv) Surface plasmon resonance (v) Non-covalent mass spectrometry Have recently opened a new facility for Biological Chemistry which includes new ITC and SPR equipment.
Notch is a single transmembrane receptor activated by ligands of the DSL family
-secretase (PS) Ligand interaction triggers a series if regulated proteolytic events wich release the intracellular domain of Notch to interacy with nuclear proteins in particular CSL
Notch plays a central role in most (all) cell fate decision during development and the differentiation of mES and hES cells Alterations in Notch activity lead to Alagile syndrome Spondylocostal dysostosis CADASIL and in many instances Cancer
T cell acute lymphoblastic leukemia (T-ALL) Grabher et al.Nature Reviews Cancer advance online publication; published online 13 April 2006 | doi:10.1038/nrc1880
Notch -secretase has other substrates Cadherin ErbB-4 ß-amyloid precursor protein (APP) Corollary: other processes might be affected -secretase inhibits Notch activation and thus has secondary effects on Notch signalling in various places e.g intestine and the immune system Corollary: one could use-secretase inhibitors to curtail CRC. However at the moment this results in all cell differentiating and an dissappearance of the epithelium through exhaustion of the niche In transgenic models induction of lymphomas through activation of Notch has a very long latency which is shortened by secondary events. The reverse is also truth e.g Ikaros-Notch connection Corollary: targetting Notch activation might not have an effect on endogenous Notch before being effective on the lymphoma.
T cell acute lymphoblastic leukemia (T-ALL) Grabher et al.Nature Reviews Cancer advance online publication; published online 13 April 2006 | doi:10.1038/nrc1880
Endocytosis Traffick Stability Transcription
-secretase has other substrates Cadherin ErbB-4 ß-amyloid precursor protein (APP) Specificity: Notch ANK only interact with Notch effectors. This allows selective interference with Notch activity. Targetting Notch ANK will not affect Notch activation but rather active Notch. It will be possible to target specific processes e.g. transcription, traffick, degradation. It Will diminish interference with normal Notch signalling. -secretase inhibits Notch activation and thus has secondary effects on Notch signalling in various places e.g intestine and the immune system In transgenic models induction of lymphomas through activation of Notch has a very long latency which is shortened by secondary events. The reverse is also truth e.g Ikaros-Notch connection Targetting Notch ANK will allow a particularly effective way of treating pathologies triggered by promiscuous transcriptional activity of Notch