Chemical Composition
Chemical Composition. My Chapter 7 Book Chapter 8. These seashells from Sanibel Island, Florida, contain the mineral calcium carbonate. The Enzo Ferrari is built of composite materials. Objective 1.

Chemical Composition
E N D
Presentation Transcript
Chemical Composition My Chapter 7 Book Chapter 8
These seashells from Sanibel Island, Florida, contain the mineral calcium carbonate.
Objective 1 • To understand the definition of Formula mass. To be able to determine the formula mass of a given compound.
one of the most useful things that a correctly written chemical formula and a periodic table can tell us is the mass of the formula 1. What unit do we use to describe mass? • grams 2. Does that fact that we will be determining the mass of groups of atoms present us with any problems? If so, how can it be solved?
3. What is Formula Mass? • the sum of all the atomic masses of all the elements in a chemical formula 4. What are the different types of chemical formulas again? • ionic compounds • molecular compounds
if you are dealing with a molecular, you will often see it referred to as molecular mass; but for all intents and purposes, they mean the same thing.
Formula Mass Ex. 1 What is the molecular mass of hydrogen gas? Hydrogen gas = H2 2 x 1.0 u = 2.0 u
Formula Mass Ex. 2 What is the molecular mass of water, dihydrogen monoxide? H20 2 H x 1.0 = 2.0u 1 O x 16.0 = 16.0u 18.0u
Formula Mass Ex. 3 What is the formula mass of aluminum nitrate? Al(NO3) 3 1 Al x 27.0 = 27.0u 3 N x 14.0 = 42.0u 9 O x 16.0 = 144.0u 213.0u
Formula Mass Ex. 4 What is the formula mass of calcium hydroxide? Ca(OH) 2 1 Ca x 40.1 = 40.1u 2 O x 16.0 = 32.0u 2 H x 1.0 = 2.0u 74.1u
Formula Mass 5. Why was the word "molecular" used for Ex's 1 and 2? Why "formula" for Ex. 3 and 4? How did you know? • Ex’s 1 and 2 were molecules thus the term molecular mass • Ex’s 3 and 4 were ionic compounds thus the term formula mass
Sulfur trioxide Potassium cyanide Lead III chromate Nitrogen IV sulfide Chromium II phosphate Phosphoric acid Copper I hydroxide Hydrofluoric acid Dinitrogen trioxide Sulfuric acid 80.1u 65.1u 762.4u 78.2u 346.0u 98.0u 80.5u 20.0u 76.0u 98.1u
Objective 2 • To understand the concept of average mass and explore how counting can be done by weighing
Suppose you worked in a candy store and people came in asking for jelly beans in quantities of 50 or 100 or 1000 beans. Could you imagine counting them out? 6. What would be a faster way of getting them what they want? 7. What if the jelly beans were exactly the same?
8. What would you do if the jelly beans were not exactly the same? 9. What would you have to do for every type of single candies in the shop? Ex. 5 If I want 100 mints and 100 jelly beans, how many grams of each would you weigh out?(Assume avg mass are 15 g for mint, 5 g for jb) • Read Chemistry in Focus on p 205 for a quiz question tomorrow
Objective 3 • To understand atomic mass and its experimental determination
How about if our job is not to sell jelly beans? • Our job is to sell atoms… • Can we use the same concept to do just that? 10. Where can you find the average masses of the different atoms? • PT--its mass number
11. How do we measure the mass of atoms? • Using amu(atomic mass units) Ex. 6 Calculate the mass, in amu, of an aluminum sample that contains 75 atoms. Ex. 7 Calculate the number of sodium atoms present in a sample that has 1172.5 amu.
Objective 4 • To understand the mole concept and Avogadro’s number. • To learn to convert among moles, mass, and number of atoms in a given sample
12. What problem do we encounter when we try to use a unit such as amu? • not very practical since most balances we use are measured in grams • 1 amu = 1.66 E -24 g
13. How could we solve this problem? • Find a way to measure atoms using grams!
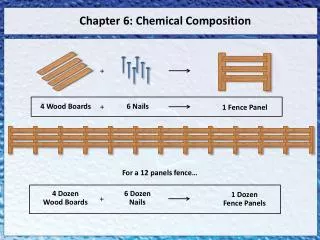
there are many words in the English language that are used to describe a certain number of things. For example: • Dozen means: 12 • Ream means: 500 • Gross means: 144
It would be impractical for a Jelly bean company to keep track of every bean that is produced.
in the same way it would be impractical to measure a few atoms at a time in grams, but, what if you had trillions of atoms?
1 atom of Carbon = 12 u-too small for grams • 100 atoms = 1200 u-still too small • 1000 atoms = 12,000u-still too small • 1,000,000 = 12,000,000-still to small • 6.02 x 1023 atoms = 12 g just the right size
14. Who came up with a mathematical way to converting amu to grams? • in the 1700's, the Italian scientist Amadeo Avogadro 15. What did Avogadro come up with? • a mole of atoms of any element equals the atomic mass of that element in grams 16. Give an example of this concept? • a mole of Carbon atoms has a mass of 12g.
A 1-mol sample of graphite (a form of carbon) weighs 12.01 g.
One-mole samples of iron (nails), iodine crystals, liquid mercury, and powdered sulfur.
Amadeo Avogadro(1776-1856) • full name, Lorenzo Romano Amadeo Carlo Avogadro • trained and worked as a lawyer • turned to science in 1800 • proposed Avogadro’s Law in 1811 and from this law came the concept of the mole
Amadeo Avogadro(1776-1856) • was never accepted in is lifetime • Law and number was finally accepted in 1860 when the Italian Stanislao Cannizzaro(1826-1910) described his experiments proving them at a tech conference in in Karlsruhe, Germany.
17. How big is a mole? • a mole of anything is 6.02 x 1023 Ex. 8 A mole of Eddie Anderson is how many? • Too Many!!! 6.02 x 1023
Ex. 9 A mole of Iron atoms is how many? How much mass do they have? • 6.02 x 1023 Fe atoms. 56.0 g Ex. 10 A mole of Oxygen molecules is how many? How about mass? • 6.02 x 1023 32.0 g
Ex. 11 A mole of Iron(III) chloride is how many? How about mass? • 6.02 x 1023 162.5 g
**Write the Think system down in order to help you convert!** When you see“Think” mole 1 formula unit N molecules N grams Formula mass L at STP 22.4 L atoms # atoms in formula x N ions#ions in formula x N
Ex. 12 1.5 moles of Silver nitrate is how many? How about mass? • 9.03 x 1023 ; 254.9g • Ex. 13 How many moles do you have when you have 40 g of C? • 3.3 mole C
Ex. 14 How many moles do you have when you have 100 g of Magnesium sulfate? • .831 mol Ex. 15 4.59 moles of Dicarbon pentoxide has how many molecules? How many grams? • 477g C2O5
here is a review of what the mole can help you to convert: 1. 1 mole of a gas is = 22.4 L at Standard Temperature and Pressure(0 C and 101.3 kPa)--this is true for any gas in the universe. If you have 1 mole of it you will have 22.4 dm3 of the gas! 2. 1 mole of a formula unit(formula of an ionic compd) or a molecule = formula mass or molecular mass of that compd
3. 1 mole of ions, molecules or formula units = 6.02 x 1023 ions, molecules, or formula units
Ex. 16 Find the number of molecules in a sample of oxygen gas with a mass of 64.0g? • 1.20 x 1024 molecules
Ex. 17 Find the volume of oxygen gas with a mass of 45.3 g at STP? Ex. 18 Find the number of ions in a 4.08 mole sample of Aluminum hydroxide?
Ex. 19 Find the mass of a sample of nitrogen gas with a volume of 36.7 L? Ex. 20 Find the number of molecules of a sample of Nitrogen dioxide gas with a volume of 453 cm3?
Now that we know how to write chemical formulas and names-well most of us anyway-let's put them to good use • there are ways of getting correctly written chemical formulas to tell important information
remember that in chemistry, Quantitative analysis is often more informative than qualitative • many mathematical relationships can be deduced by using chemical formulas and chemical equations, this study into the quantitative nature of chemical formulas is called stoichiometry
Objective 5 If given the percentage composition or the mass amounts of each element, be able to determine the empirical formula. If given the molecular mass, be able to determine the molecular formula as well.
Percentage Composition • you can do all sorts of neat stuff with a correct chemical formulas 18. What is percentage composition? • percentage by mass of each of the elements in a cmpd
19. What do chemists do with percentage composition of a cmpd? • Determine how much of a particular element can be found in a compound. Very useful in the mining industry where the determination of the % composition of the ore will decide whether it is worth mining at that site. • It can also help us determine the chemical formula of an unknown compound
Ex. 21 What are the percentage compositions of the following: carbon dioxide CO2 1 C x 12.0 = 12.0u 2 O x 16.0 = 32.0u 44.0 12.0 /44.0 x 100 = 27.3% C 32.0 / 44.0 x 100 = 72.7% O