Chemistry Solutions and Precipitates: Understanding Mixtures & Reactions
100 likes | 125 Vues
Explore the world of chemistry, from mixtures composition to disappearing orange reactions. Learn about solutions, precipitates, and ionic species. Discover Redox reactions and the mysteries behind color changes.

Chemistry Solutions and Precipitates: Understanding Mixtures & Reactions
E N D
Presentation Transcript
SOLUTIONS AND PRECIPITATES • What mixes, what doesn’t and Why
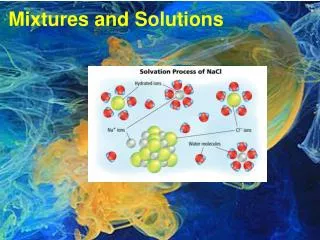
MIXTURES: 1 + 1 = 2or Does It? • Add 50 mL of H2O to 50 mL of H2O • ⃗ 100 mL Mixture • Add 50 mL of H2O to 50 mL of MeOH • i.e Water + Methanol (Methyl Alcohol) • ⃗ ?? mL Mixture. WHY?
IMAGING INTERNAL X-RAYS • Need DENSE material to Block X-Rays (BaSO4) • 2BaCl + Na2SO4→Ba2SO4 + 2NaCl • Note: Ba2SO4 is VERY INSOLUBLE in WATER
DISAPPEARING ORANG E REACTION: Now You See It, Now You Don’t ! • Solution A – Potassium Iodide (KI) in H2O • Solution B – Mercuric Chloride (HgCl2) in H2O
DISAPPEARING ORANG E REACTION: Now You See It, Now You Don’t ! • Questions: • Is this a REDOX Reaction? • How can you explain the disappearance of the Orange Color?
DISAPPEARING ORANG E REACTION: Now You See It, Now You Don’t ! • 1.Hg2+ + 2I-⇒HgI2 (Orange) • 2. HgI2 + 2I-⇒ HgI42- (Colorless)
MIXTURES: 1 + 1 = 2or Does It? • Add 50 mL of H2O to 50 mL of H2O • ⃗ 100 mL Mixture • Add 50 mL of H2O to 50 mL of MeOH • i.e Water + Methanol (Methyl Alcohol) • ⃗ ?? mL Mixture.