Electron shells
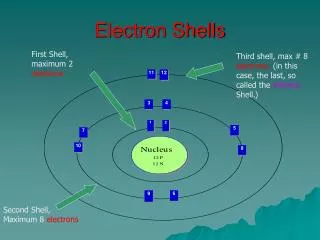
Electron shells . Electrons move rapidly around the nucleus in areas called shells. Each shell can hold up to 8 electrons except the first shell which can only hold 2 electrons The number of the period (row) an element is in, is the same as the number of shells it has.

Electron shells
E N D
Presentation Transcript
Electron shells Electrons move rapidly around the nucleus in areas called shells.
Each shell can hold up to 8 electrons except the first shell which can only hold 2 electrons The number of the period (row) an element is in, is the same as the number of shells it has
The first shell needs to be full before the second shell gets any electrons. • The last, or outer, shell is called the VALENCE SHELL. • The electrons in the valence shell are called valence electrons
Electron Shell Diagrams (Also called Bohr diagrams after the man who started them.)
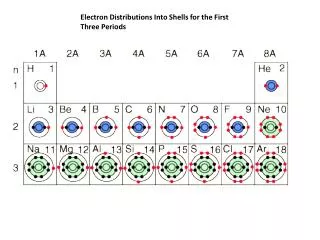
The atomic number tells you the number of protons an element has. • In its natural state, each element has the same number of electrons as protons. • So: We use the atomic number to know how many electrons we need to draw. Then we start filling up shells from the inside out.
Reminders • We write the element symbol in the center. • The first shell is full at 2. • Every other shell is full at 8. • We always draw 4 single electrons before we start to pair them up.