Chemistry 11: Typical Elements
Chemistry 11: Typical Elements. Christopher Chui. Hydrogen. Members of a family or group tend to react similarly Hydrogen is usually considered as a family by itself A hydrogen atom can react to lose 1 electron H +

Chemistry 11: Typical Elements
E N D
Presentation Transcript
Chemistry 11: Typical Elements Christopher Chui Typical Elements -- C. Chui
Hydrogen • Members of a family or group tend to react similarly • Hydrogen is usually considered as a family by itself • A hydrogen atom can react to lose 1 electronH+ • A hydrogen atom can also share its electron with nonmetals, such as HCl or H2O • The 3rd way is that a hydrogen atom gains 1 electron to become a hydride ion, H- • A 4th way is to form bridges, such as boron hydride, B5H9 • A 5th way is to add hydrogen to the double bonds of carbon in organic chemistry Typical Elements -- C. Chui
Alkali Metals • The alkali metal atoms increase in size with increasing atomic number: Li, Na, K, Rb, Cs, Fr • As the atomic numbers increase: atoms become larger; outer electrons are farther from nucleus; lower level electrons shield effect of larger nucleus; outer electrons are held less tightly; and atoms become more active • Sodium compounds are used as catalysts and in the production of paper, glass, detergents, soaps, pigments, and petroleum products • A catalyst is a substance that speeds up a reaction • Lithium is used to make batteries. It forms the 1+ ion • Alkali metals form binary compounds with most nonmetals Typical Elements -- C. Chui
Alkali Earth Metals • Alkaline earth metals form the 2+ ion • Most group 2 (IIA) compounds are soluble in water • The metals (Ca, Sr, Ba, Ra) will displace hydrogen from water and other compounds • Calcium oxide (lime) is used to make steel, cement, and heat-resistant bricks • Calcium chloride is used in making paper and pulp • Calcium is important in the structure of bones and teeth. Calcium ions are necessary for the contraction of muscles. Calcium ions also take part in the reaction that cause blood to clot Typical Elements -- C. Chui
Aluminum, and Carbon • Aluminum is the most plentiful metal in Earth’s crust • Aluminum (Gp 13, IIIA) tends to share its 3 outer electrons in forming compounds • Aluminum sulfate is used in water purification, paper manufacture, and fabric dyeing • Carbon (Gp 14, IVA) reacts by sharing 4 electrons • Carbon forms long chains by catenation • Diamond and graphite are 2 allotropes of carbon • Carbon dioxide is used in refrigeration, beverages, and in making other chemicals • Small crystals of dry ice are used in cloud-seeding • Silicon is the 2nd most plentiful element in Earth’s crust • Silicon bonds by sharing electrons • Tin: 4+ is more stable than 2+; and lead: 2+ is more stable than 4+. Typical Elements -- C. Chui
Nitrogen and Phosphorus • N2 and P4 are found in Gp 15 (VA) • N2 occurs in oxidation states from 3- through 5+ • P4 occurs only in 3-, 0, 3+, and 5+ • N2 gas is very stable, but its compounds are not • TNT and dynamite are made with N2 • Liquid N2 has extensive industrial use • Ammonium nitrate is used as fertilizer or explosives • P4 molecules stack in different ways to form allotropes • Phosphoric acid is used to make fertilizer and other applications • Organic compounds of phosphorus are vital to living organisms Typical Elements -- C. Chui
Oxygen Family • Oxygen is the most plentiful element in Earth’s crust • Oxygen reacts by gaining 2 electrons or by sharing electrons • It loses electrons with metals to form ionic oxides. Ionic oxides react with H2O to form basic solutions • It shares electrons with nonmetals. The nonmetallic oxides react with H2O to form acidic solutions. This dual behavior is called amphoteric • O2 has several allotropes. Their properties are similar to that of sulphur • S can form long chain ions such as S62- • Other members of the group: Se, Te, and Po Typical Elements -- C. Chui
Halogens • Group 17 (VIIA) contains F, Cl, Br, I, and As • Halogens react by forming negative ions or by sharing electrons • As the atomic numbers increase: atoms become larger; outer electrons are farther from nucleus; lower level electrons shield effect of larger nucleus; nucleus has less attraction for electrons of other atoms; and atoms become less active • F is the most reactive chemical element • Cl forms chlorides readily. The primary use of Cl is to make other chemicals • Halogens form compounds such as CIF, ClF5, BrF5, IF5, IF7, BrCl, and ICl3 Typical Elements -- C. Chui
Noble Gases • Group 18 (VIIIA) contains very stable gases, because all outer electrons are filled • Chemically, noble gases are not inert • Xe, Kr, and Rn compounds have been made • Xenon hexafluoroplatinate (XePtF6) was first made in 1962 • Ar is used to fill light bulbs to protect the filament • Ne, Kr, and Xe are used to fill brightly colored gas discharge tubes for advertising Typical Elements -- C. Chui
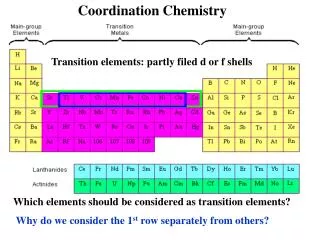
Transition Metals-1 • Transition elements have electrons filling d sublevels • Transition metals have many uses. Au, Ag, Cu, Ni, and Pt are coinage metals. Catalytic converters contain Pt, Pa, and Rh. Os is used to harden pen points. Co-60 is used to treat cancer. Mo is used in spark plugs • Transition elements, used alone or as alloys, are our principal structural metals: Ti through Zn. TiO2 is used in paints. MnO2 is used in batteries. Fe and Cr oxides are used in audiotapes and videotapes Typical Elements -- C. Chui
Transition Metals-2 • Cr is important because it resists corrosion. It is used in stainless steel and chromeplate regular steel • Cr reacts by losing 3 electrons to form the Cr 3+ ion • Cr also forms the 6+ states in CrO42- , and Cr2O72-Many Cr compounds are colored. Small amount of Cr are found in crystals of rubies and emeralds • Zn behaves different because of its full d sublevel. It has one oxidation state, 2+ • Metallic Zn is corrosion resistant. It is used to coat Fe. This process is called galvanizing • Zn is essential to proper body function Typical Elements -- C. Chui
Neodymium (Lanthanoid), Curium (Actinoid) • Nd, as all lanthanoids, shows 3+ as the most stable oxidation state • Nd metal is soft and very reactive. It forms alloys with unusual conductivity and magnetic properties. Nd2O3 is used in glass filters and in lasers • Curium (Cm) has a half-full f sublevel. It forms 3+ oxidation compounds. Its solution is pale yellow • Cm is a slivery, hard metal of medium density and high melting point. It does not occur in nature. It is produced by slow neutron bombardment of Pu • Cm is reactive and highly toxic to humans • Cm is used as energy source in satellites Typical Elements -- C. Chui