Periodic Trends in the Modern Periodic Table
190 likes | 214 Vues
Learn about the development and organization of the periodic table by Mendeleev and Moseley, along with key trends such as atomic size, ion size, electronegativity, and ionization energy. Understand the foundational principles behind how elements are classified and their properties.

Periodic Trends in the Modern Periodic Table
E N D
Presentation Transcript
Chapter 6: Periodic Trends
Development of the Periodic Table • Dmitri Mendeleev designed the first periodic table in 1869 by grouping elements with similar chemical & physical properties in rows according to Atomic mass • Henry Moseleyrearranged the table in 1913 according to atomic number
The Modern Periodic Table Organized by groups (columns) and periods (rows)
Classifying Elements by Electron Configurations • Representative Elements- Outermost s and p are filling • Alkali metals – Outermost s has 1 e- • Alkaline earth metals – Outermost s is full • Halogens – Outermost s is full, outermost p has 5 e- • Noble Gases- Outermost s and p are totally full • Transition Metals -Outermost s is full and d is filling or full • Inner Transition Metals- Outermost s is full and f is filling or full
Periodic Trends As you move across the periodic table, you keep periodically running across the same properties... • The location of an element on the Periodic Table gives a lot of information about key properties of that element compared to the other elements Hence the name periodic table.
Trend #1: Atomic Size • An atoms size is determined by its atomic radius • Atomic radius is defined as half the distance between the nuclei of two like atoms r
Atomic Size Trend • Going down a group, atoms get bigger because the number of energy levels increases. • Going across a period, atoms get smaller. No new energy levels are added, but more protons and electrons are. The increase in protons and electrons result in a greater pull towards the nucleus. (much like magnets)
Trend #2: Atomic Size of IONS • Cations (positive ions) are always smaller than their parent atom because they lose electrons and an energy level. A strong attraction forms between the electrons left over and the nucleus. • Anions (negative ions) are always larger than their parent atom. Gaining electrons causes less attraction to the nucleus – the new electrons aren’t as attracted and are free to move around!
Answer this . . . Which is larger: S or S-2. Why? • S-2 is larger – Gaining electrons causes less of an attraction to the nucleus – the new electrons are free to move around. Which is smaller: Fe or Fe+4. Why? • Fe+4 is smaller – Losing electrons causes more of an attraction to the nucleus – the remaining electrons are pulled in tighter. Which is smaller: Na+ or Al3+? Why • They both lose an energy level BUT Al3+loses MORE e-, nucleus pulls in the remaining electrons due to the more drastic change.. Which is smaller: Be2+ or Na+. Why? • They both lose an energy level BUT Be2+ loses MORE e- & goes down to a smaller energy level. A neutron walks into a ‘restaurant’ and says, "Hey bartender give me a drink."The bartender gives him one and says, “No charge for you" An atom walks into a ‘restaurant’and proclaims, "Hey! Somebody just stole one of my electrons!"The bartender says, "Are you sure?“ The atom replies, "Yes - I'm positive!"
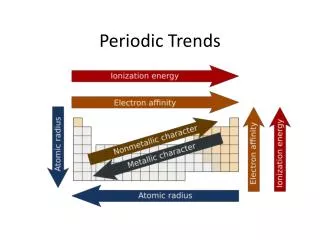
Trend #3: Electronegativity ELECTRONEGATIVITY - the ability for an element to attract an electron Going down a group, electronegativity decreases because the added energy levels ‘shield’ the power of the nucleus to attract electrons Going across a period, electronegativity increases because nuclear charge increases - the closer an atom is to having a full outer shell, the greater the desire to completely fill that shell by gaining electrons.
Which element would be the most electronegative? Why? • Fluorine - It is located at a low energy level with a high number of electrons and protons. There is a strong attraction between the electrons and the nucleus.It wants an electron BADLY!
How does ‘Shielding’ work? • SHIELDING EFFECT: The process of the inner electrons shielding (repelling) the outer electrons - it causes the outer electrons to be less attracted to the nucleus. • Shielding increases as you go down the periodic table because more electrons in more energy levels are added • Shielding remains constant as you go across because all the electrons in a period are in the same energy level.
Why aren’t noble gases assigned electronegativity numbers? • They are happy the way they are – they don’t need any more electrons
Trend #4: Ionization Energy IONIZATION ENERGY- The energy needed to REMOVE an electron from the outside shell. Going down a group, ionization energy decreases because more energy levels are added. This ‘shielding’ causes the electrons to be less attracted to the nucleus - little energy is needed to remove them. Going across a period, ionization energy increases because the electrons are closer to the nucleus. More energy is needed to remove them the closer they are to the nucleus.
First vs. Second • First ionization energy is the energy required to remove the first electron • Second ionization energy is the energy required to remove the second electron (and so forth) • First ionization energy is always smaller (compared to the second or third ionization energy) because each successive electron removed is closer to the nucleus and strongly attracted to it.
Trend #5: Metallic PropertiesFirst off, what’s meant by ‘metallic’?
Metallic properties increase as you go down a group, and decrease as you go across a period. X Fr is the most metallic!