Chain Growth Polymerization (Addition-Polymerization)
450 likes | 1.34k Vues
Chain Growth Polymerization (Addition-Polymerization). Lecture No.3. Radical Chain Polymerization: “Molecule ‘Empire Building’ by ‘Radical’ Groups”. Chain-Growth Polymerization (Addition) Processes. 1. Free radical Initiation Processes. 2. Cationically Initiated Processes.

Chain Growth Polymerization (Addition-Polymerization)
E N D
Presentation Transcript
Chain Growth Polymerization(Addition-Polymerization) • Lecture No.3
Radical Chain Polymerization: “Molecule ‘Empire Building’ by ‘Radical’ Groups” Chain-Growth Polymerization (Addition) Processes • 1. Free radical Initiation Processes • 2. Cationically Initiated Processes • 3. Anionically Initiated Processes • 4. Group Transfer Polymerization • 5. Coordination Polymerization
Characteristics of Chain-Growth Polymerization 1. Only growth reaction adds repeating units one at a time to the chain 2. Monomer concentration decreases steadily throughout the reaction 3. High Molecular weight polymer is formed at once; polymer molecular weight changes little throughout the reaction. 4. Long reaction times give high yields but affect molecular weight little. 5. Reaction mixture contains only monomer, high polymer, and about 10-8 part of growing chains.
The Chemistry of Free Radical Polymerization Radical Generation R R 2 R - Initiator Radicals Initiation R C C R C C + Monomers Propagation R C C C C R C C C + Termination C C C R R C C + R C C C C C R Polymer
Free Radical Polymerization Mechanisms 1. Overview –Free radical polymerization processes involve at least three mechanistic steps. • A. Initiation • 1. Radical Formation (Generation) • D In In In In + h v , etc. 2. Initiation In M In M +
B. Propagation In-M1. + M2 In-M1M2. In-M1M2. + M3 In-M1M2M3. In-M1M2M3…MX. + MY In-M1M2M3…MXMY.
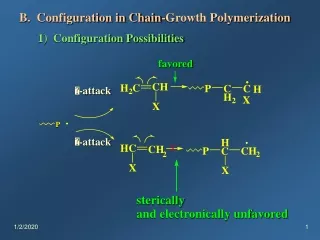
C. Termination 1) Radical Coupling (Combination) In-MX. + .MY-In In-MX-MY-In In In In In + 2) Disproportionation (-hydrogen transfer) H H H H In M In M C C C C + y x H H H H CH In M M In H C CH CH + x y 3 2 2
D. Chain Transfer (sometimes)– An atom is transferred to the growing chain, terminating the chain growth and starting a new chain. Chain Transfer to Chain Transfer Agent: R P H R P + x + x Chain Transfer to Monomer: Px. + H2C=CH-(C=O)OR Chain Transfer to Polymer: Causes Branching H P P P P + x + y x y
E. Inhibition and Retardation– a retarder is a substance that can react with a radical to form products incapable of reacting with monomer. An inhibitor is a retarder which completely stops or “inhibits” polymerization. 2. Monomers that are susceptible to free radical addition A. Vinyl Monomers H C CHX H C CH Cl 2 2 Vinyl chloride H F H X H F H Y Vinylidene fluoride
B. Allyl Monomers Cl X Allyl Chloride C. Ester Monomers 1) Acrylates OR OH O O Acrylic Acid Acrylate Esters
2) Methacrylates O O OH OR Methacrylic Acid Methacrylate Esters 3) Vinyl Esters O Vinyl Acetate O D. Amide Monomers O O NH NH 2 2 Acrylamide Methacrylamide
3. Monomers that are not susceptible to Free Radical Addition A. 1,2-a-olefins (Polymerize to oils only) x B. Vinyl ethers R O O methyl vinyl ether C. 1,2-disubstituted Ethylenes Cl Cl 1,2-dichloroethylene H H