The use of Cochrane breast cancer reviews by guideline developers and Cochrane (public) users
150 likes | 341 Vues
The use of Cochrane breast cancer reviews by guideline developers and Cochrane (public) users. Cochrane Breast Cancer Group, NHMRC Clinical Trials Centre. ML Willson, D O’Connell, A Goodwin, N Wilcken. Setting the scene. A scoping exercise

The use of Cochrane breast cancer reviews by guideline developers and Cochrane (public) users
E N D
Presentation Transcript
The use of Cochrane breast cancer reviews by guideline developers and Cochrane (public) users Cochrane Breast Cancer Group, NHMRC Clinical Trials Centre ML Willson, D O’Connell, A Goodwin, N Wilcken
Setting the scene • A scoping exercise • Increasing volume of trial data available (Bastian et al, PLoS Medicine 2010) • Utility of systematic reviews: • can guide treatment decisions • part of the research evidence component of clinical practice guidelines • Aim: • assess Cochrane review usage by clinical guidelines and our readers
Cochrane Breast Cancer Group: producers of Cochrane (systematic) Reviews • Established in 1996 • Search for evidence, evaluate its quality and publish results as Cochrane Reviews • Published in an online database and available via The Cochrane Library • Reviews known to be methodologically rigorous • Over 88 Protocols, Reviews and Titles • Sometimes integrated into clinical practice guidelines
Cochrane Breast Cancer Group: Review portfolio • Topics are decided upon by an international, multidisciplinary Editorial Board
Cochrane Breast Cancer Group: metrics • Publishers provided a short list of 8 ‘most cited’ + top 10 ‘most accessed’ • 13 of the 18 ‘most cited/accessed’ Review topics covered: • Prevention, early detection, familial breast cancer, allied health (ie exercise), psychosocial care, complementary therapy, lymphoedema • 5 of the 18 covered treatments • Chemo- and targeted therapy, radiation and surgery
Specific aims • Assess the uptake of Cochrane breast cancer Reviews in clinical practice guidelines • Compare ‘most cited/accessed’ Reviews to those used in guidelines
Methods: guideline selection & basic metrics • Selected 7 key clinical guideline developers or consensus panels: • Advanced Breast Cancer Consensus (ABC1) • American Society of Clinical Oncology (ASCO) • Cancer Australia (CA) • Central European Cooperative Oncology Group (CECOG) • European Society for Medical Oncology (ESMO) • National Institute for Health and Care Excellence (NICE) • Scottish Intercollegiate Guideline Network (SIGN) • Published from 2001 to March 2014 • For each guideline, recorded: • Number and title of Cochrane Review citations • Scope of guideline • Categorised Cochrane Review usage for each guideline developer • Compared Reviews in guidelines to ‘most cited/accessed’ Reviews
Results: no. and scope of guidelines • 28 guidelines included, published July 2001 to July 2013
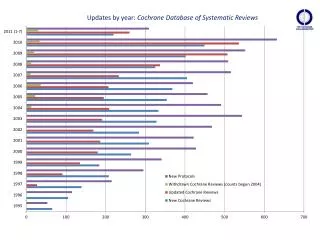
Results: Cochrane Review usage over time Cumulative no. of guidelines + Cochrane Reviews in guidelines
Results: summary • An increase in Cochrane review uptake in guidelines over time • Variation in Cochrane review citations by guideline developers • An overlap with Reviews used in guidelines and ‘most cited/accessed’ Reviews • 10 of 18 ‘most cited/accessed’ Reviews were used once or more than once • Slight disconnect • 8 of 18 ‘most cited/accessed’ Reviews were not used • i.e. peripheral topics
Limits, discussion, future implications • A preliminary assessment; small sample (n=28) • Room to improve communications with guideline developers about upcoming guideline topics • Identified some gaps in our Topics list that will need to be addressed • ‘Most cited/read’ reviews tended to be on peripheral topics (e.g. exercise) • Results provide a useful baseline for monitoring the use of our Cochrane reviews
Acknowledgements • A/Prof Nicholas Wilcken (Joint Co-ordinating Editor) • Dr Annabel Goodwin (Joint Co-ordinating Editor) • Prof Dianne O’Connell (Statistical Editor) • Cochrane Breast Cancer Group funders: • National Health and Medical Research Council (NHMRC) • National Breast Cancer Foundation (Australia) • Supported by: • NHMRC Clinical Trials Centre, University of Sydney